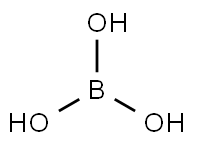
Boric acid CAS#10043-35-3
CAS Number: 10043-35-3
Chemical Formula: MnO2
Synonyms:
Manganese(IV) oxide, -100 mesh, 99+%
Manganese(IV) oxide, average particle size 2 μm, 99+%
Manganese(IV) oxide, powder, 80-85%
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: White Powder
Boric acid CAS#10043-35-3
Boric acid is a crystalline compound formed by the dissolution of boron trioxide in water. It is an inorganic acid, primarily consisting of tetraboric acid (H₂B₄O₇, also known as pyroboric acid), metaboric acid (HBO₂)n, and orthoboric acid (H₃BO₃, also known as orthoboric acid). Orthoboric acid has the highest water content and is the most stable. Orthoboric acid, also simply called boric acid, appears as a white, powdery crystalline substance or triclinic scaly crystals with a glossy appearance. It has a slightly bitter taste and a slippery feel upon contact with skin. It is odorless and soluble in water, ethanol, glycerin, ethers, and essential oils. Its aqueous solution is weakly acidic. When heated to 70-100℃, orthoboric acid gradually dehydrates to metaboric acid (Chemicalbook), to pyroboric acid at 150-160℃, and to boric anhydride (B₂O₃) at 300℃. Boric acid is toxic to humans. Solubility in water: 2.77 at 0℃, 3.65 at 10℃, 4.87 at 20℃, 5.74 at 25℃, 6.77 at 30℃, 8.90 at 40℃, 11.39 at 50℃, 14.89 at 60℃, 23.54 at 80℃, and 38.00 at 100℃. The aqueous solution is weakly acidic, making it a typical Lewis acid. Its solubility in water is increased in the presence of hydrochloric acid, citric acid, or tartaric acid. Therefore, it can be purified by recrystallization.
Application of Boric acid
Boric acid is used as a pH adjuster, disinfectant, and antibacterial preservative. It is also used in capacitor manufacturing and the electronic component industry, as a high-purity analytical reagent, in pharmaceutical disinfection and preservation, and in the preparation of washing solutions for exposed photosensitive materials.
It is used in the glass, enamel, ceramics, pharmaceutical, metallurgical, leather, dye, pesticide, fertilizer, and textile industries.
It is used as a chromatographic analytical reagent and in the preparation of buffers. It is widely used in the glass industry (optical glass, acid-resistant glass, heat-resistant glass, and glass fiber for insulation), improving the heat resistance and transparency of glass products, increasing mechanical strength, and shortening melting time. In the enamel and ceramics industry, it is used to enhance the gloss and durability of enamel products and is also a component of glazes and pigments. In the metallurgical industry, it is used as an additive and co-solvent; boron steel, in particular, has high hardness and good rollability and can replace nickel steel. Boric acid has preservative properties and can be used as a chemical book preservative, such as for wood preservation. Boric acid is used in metal welding, leather, photography, and the manufacture of dyes, heat-resistant and fire-retardant fabrics, artificial gemstones, capacitors, and cosmetics. It can also be used as an insecticide and catalyst. In agriculture, boron-containing micronutrient fertilizers are effective for many crops, improving crop quality and increasing yield. Boric acid is also one of the basic raw materials for producing other boron compounds, and the boron compounds produced from it are widely used in defense and other industrial sectors and research institutions.
Fact Factory and Equipment Show