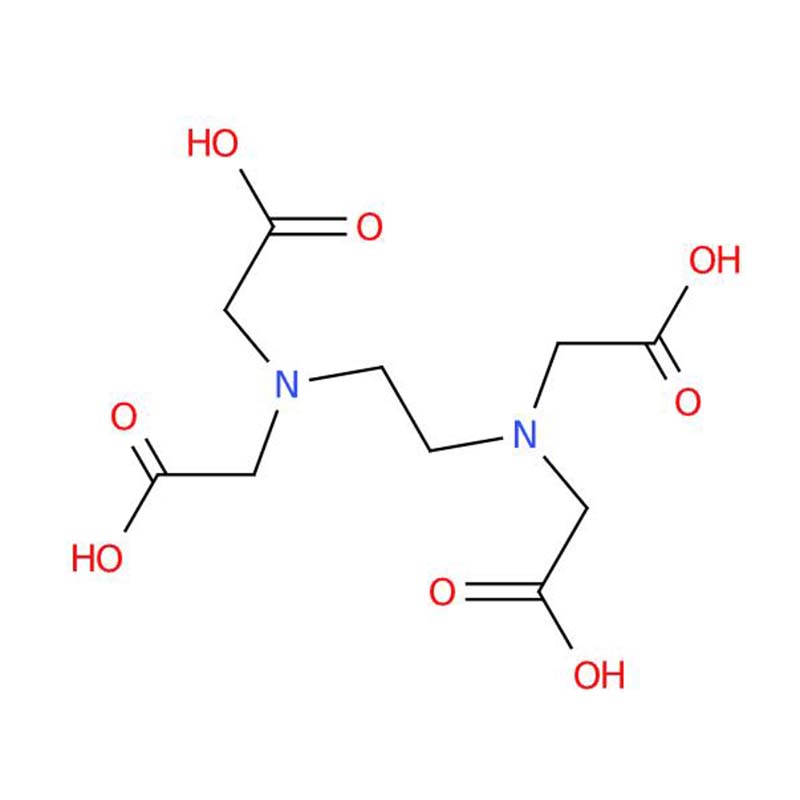
Ethylenediaminetetraacetic acid CAS:60-00-4
Ethylenediaminetetraacetic acid, abbreviated as EDTA, is an organic substance with the molecular formula C10H16N2O8. Under standard temperature and pressure conditions, it appears as a white crystalline powder. As a typical chelating ligand, EDTA is capable of forming stable complexes with divalent metal ions including magnesium, calcium, manganese and iron ions.
Given that many nucleases and certain proteases rely on magnesium ions to maintain their catalytic activity, EDTA is widely employed to inhibit these enzymes. Additionally, it can be used to counteract the inhibitory influence exerted by heavy metal ions on various enzyme systems.
Ethylenediaminetetraacetic acid serves as the acidic precursor of edetate and functions as an effective chelating compound, featuring the ability to resist hypercalcemia and inhibit blood coagulation. It can bind with calcium ions and heavy metal ions to generate soluble and highly stable coordination complexes. This substance is widely applied as an anticoagulant in blood specimen collection and is also adopted in clinical interventions for lead poisoning.
| Density | 1.6±0.1 g/cm3 |
| Boiling point | 614.2±55.0 °C at 760 mmHg |
| Melting point | 250 °C (dec.)(lit.) |
| Molecular formula | C10H16N2O8 |
| Molecular weight | 292.243 |
| Flash point | 325.2±31.5 °C |
| Precise mass | 292.090668 |
| PSA | 155.68 |
| LogP | -0.43 |
| InChIKey | KCXVZYZYPLLWCC-UHFFFAOYSA-N |
| SMILES | O=C(O)CN(CCN(CC(=O)O)CC(=O)O)CC(=O)O |
| Appearance characteristics | White crystals or powder |
| Steam pressure | 0.0±3.8 mmHg at 25°C |
| Refractive index | 1.58 |
| Storage conditions | 1. Store in a cool and dry place. |
| 2. Do not mix with alkaline chemicals. | |
| Stability | 1. The solubility in water is 0.5g/L (at 25℃). A decarboxylation reaction will occur when heated to 150℃. |
| 2. It should be avoided from being ingested, inhaled or coming into contact with the skin or eyes. | |
| 3. It can form stable complexes with various metal ions and is commonly used as a complexing agent in electroplating and chemical plating. | |
| Water solubility | 0.5 g/L (25 ºC) |
| Molecular structure | 1. Molecular refractive index: 62.07 |
| 2. Molar volume (cm³/mol): 186.6 | |
| 3. Isothermal volume (90.2K): 568.5 | |
| 4. Surface tension (dyne/cm): 86.1 | |
| 5. Polarizability (10⁻²⁴ cm³): 24.60 |