AcetaminophenCAS:103-90-2
Therapeutic Efficacy in Pain and Fever Management
Paracetamol (also known as acetaminophen or 4-acetamidophenol) functions as a widely prescribed analgesic-antipyretic agent, celebrated for its robust ability to mitigate mild to moderate pain and reduce febrile symptoms . Its dual-action mechanism—targeting pain-signaling pathways and regulating the hypothalamic temperature set point—has solidified its status as a cornerstone ingredient in over-the-counter pain relief formulations worldwide .
Stability Profile
The chemical integrity of this compound remains uncompromised at temperatures not exceeding 45°C, with optimal stability observed under controlled storage conditions of 20–25°C (68–77°F) . When preserved in cool, dry environments and protected from light and moisture, paracetamol retains its pharmaceutical potency and therapeutic reliability over extended periods, minimizing degradation risks associated with humidity or extreme temperatures .
Solubility Characteristics
Paracetamol exhibits enhanced solubility in heated aqueous solutions (14 mg/mL at 20°C, increasing significantly with temperature) and ethanolic solvents, while demonstrating limited solubility in cold water (7.21 g/kg at 0°C) . This solubility profile facilitates rapid dissolution upon administration, ensuring high bioavailability (peak plasma concentrations achieved within 0.5–2 hours) and supporting diverse formulation approaches—including tablets, syrups, and injectables .
Established Clinical Legacy
Chemical Classification and Nomenclature
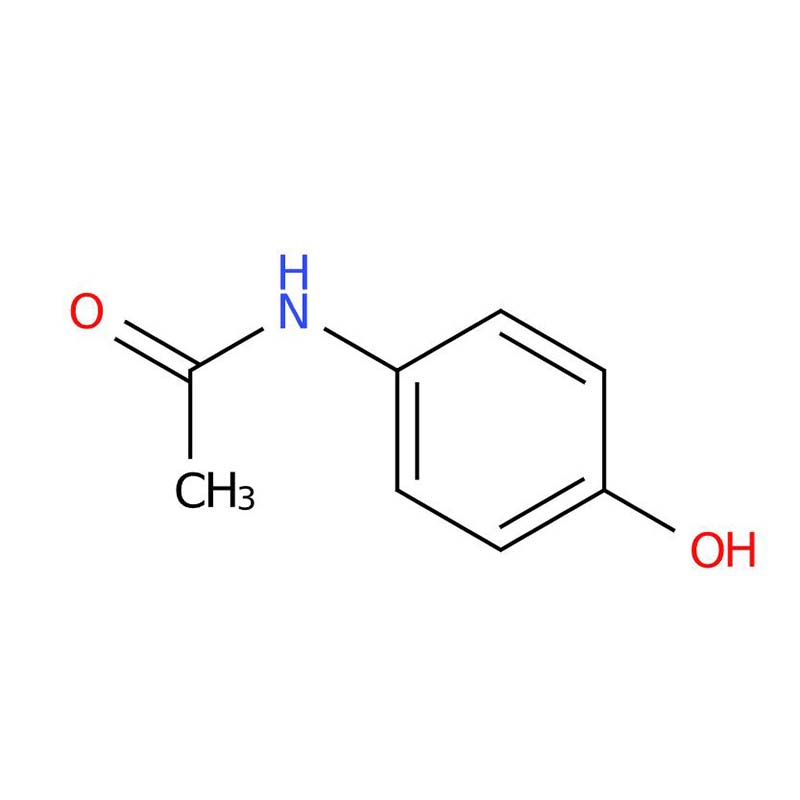
Acetaminophen, with the systematic IUPAC name N-(4-hydroxyphenyl)acetamide and the widely recognized commercial name paracetamol, belongs to the class of antipyretic-analgesic agents derived from acetanilide . Distinct from non-steroidal anti-inflammatory drugs (NSAIDs) due to its unique pharmacological mechanism and safety profile, this compound occupies an independent category in clinical pharmacology .
Historical Milestones in Synthesis and Application
The synthesis of this compound was first accomplished by Harmon Northrop Morse, a professor at Johns Hopkins University, in 1878 through the reaction of p-nitrophenol and glacial acetic acid under tin catalysis . However, its therapeutic potential remained unrecognized for over a decade until 1893, when German pharmacologist Von Mering documented its initial clinical application, laying the foundation for its subsequent medical use .
Notably, the journey of acetaminophen from laboratory synthesis to global widespread use was shaped by key scientific discoveries—including the identification of its role as the active metabolite of acetanilide in the 1940s, which clarified its therapeutic mechanism and spurred its clinical promotion .
Melting point | 168-172 °C(lit.) |
Boiling point | 273.17°C (rough estimate) |
density | 1,293 g/cm3 |
vapor pressure | 0.008Pa at 25℃ |
refractive index | 1.5810 (rough estimate) |
Fp | 11 °C |
storage temp. | Inert atmosphere,Room Temperature |
solubility | ethanol: soluble0.5M, clear, colorless |
form | Crystals or Crystalline Powder |
pka | 9.86±0.13(Predicted) |
color | White |
PH Range | 5.5 - 6.5 (H?O, 20 °C) (saturated solution) |
PH | 5.5-6.5 (H2O, 20℃)(saturated solution) |
Odor | odorless |
explosive limit | 15%(V) |
Water Solubility | 14 g/L (20 ºC) |
Merck | 14,47 |
BRN | 2208089 |
BCS Class | 3,4 |
InChIKey | RZVAJINKPMORJF-UHFFFAOYSA-N |
LogP | 1.098 at 25℃ |
CAS DataBase Reference | 103-90-2(CAS DataBase Reference) |
NIST Chemistry Reference | Acetaminophen(103-90-2) |
IARC | 3 (Vol. 50, 73) 1999 |
EPA Substance Registry System | Acetaminophen (103-90-2) |
Safety Information | |
Hazard Codes | Xn,T,F |
Risk Statements | 22-36/37/38-52/53-36/38-40-39/23/24/25-23/24/25-11 |
Safety Statements | 26-36-61-37/39-22-45-36/37-16-7 |
RIDADR | UN 3077 9/PG III |
WGK Germany | 1 |
RTECS | AE4200000 |
Autoignition Temperature | 540 °C |
TSCA | Yes |
HazardClass | 9 |
PackingGroup | III |
HS Code | 29242930 |
Hazardous Substances Data | 103-90-2(Hazardous Substances Data) |
Toxicity | LD50 in mice (mg/kg): 338 orally (Starmer), 500 i.p. (Dahlin, Nelson |
Chemical Stability and Degradation Characteristics
Acetaminophen retains its chemical stability when stored at temperatures not exceeding 45°C, preserving its structural integrity under recommended thermal conditions . Nevertheless, exposure to humid environments triggers hydrolytic breakdown of the compound, leading to the formation of p-aminophenol as the primary degradation product . This intermediate metabolite further undergoes oxidative conversion through subsequent chemical reactions, initiating a visible chromatic evolution—progressing from a faint pinkish tint to a deep brown shade, and eventually developing dark black pigmentation as degradation advances . Such color changes serve as a direct visual indicator of compromised product quality, underscoring the necessity for storage in cool, moisture-free (desiccated) environments to inhibit hydrolytic and oxidative degradation pathways .
Physical Appearance and Key Physicochemical Traits