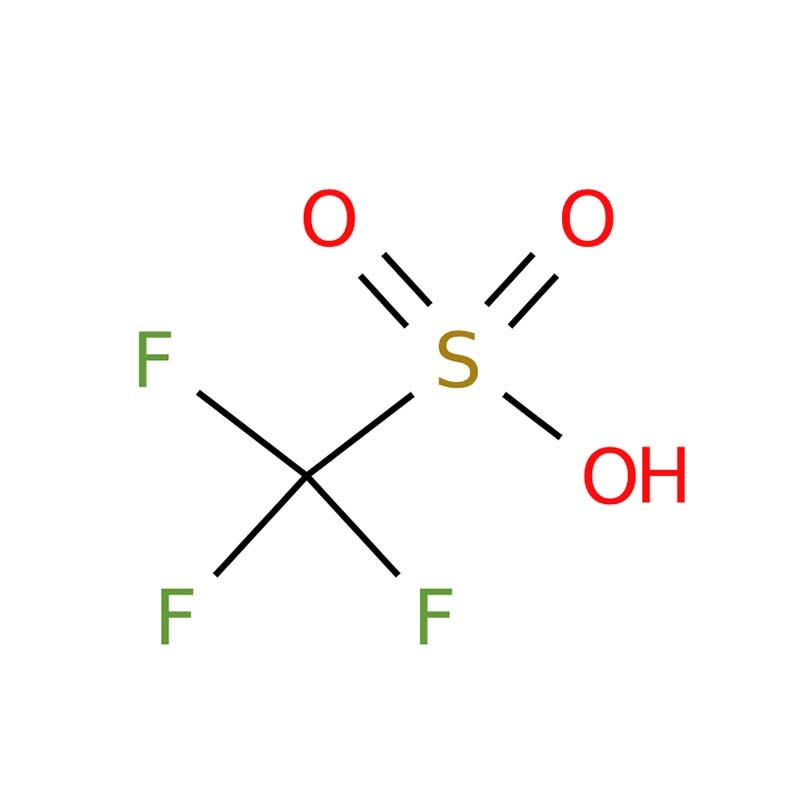
Trifluoromethanesulfonic Acid (TfOH) CAS No.: 1493-13-6
Trifluoromethanesulfonic acid (commonly known as triflic acid, TfOH, CAS 1493-13-6) is a colorless to slightly brown fuming liquid, recognized as one of the strongest available monoprotic organic acids with a pKa of approximately -14.7 . It is miscible with water and polar organic solvents, though dissolution is highly exothermic and requires careful handling due to its corrosive, hygroscopic nature . Unlike sulfuric or nitric acids, triflic acid is valued in organic synthesis for its thermal stability and non-oxidizing characteristics, with its conjugate base (triflate) being weakly nucleophilic . It serves as an extremely potent acid catalyst for reactions including Friedel-Crafts acylations, cationic polymerizations, and glycosylation, while also being used in the electronics industry as an electrolyte component and as a key precursor for synthesizing triflate derivatives and ionic liquids
1. Overview
Trifluoromethanesulfonic acid is one of the strongest organic acids (pKa ≈ -14). It appears as a colorless to pale yellow fuming liquid. Unlike traditional superacids, it is thermally stable and non-oxidizing. Widely used in catalysis, battery electrolytes, and pharmaceutical synthesis due to its high acidity and excellent solubility in organic solvents.
2. Key Features
Super Strong Acidity: Approximately 1000x stronger than sulfuric acid (H₂SO₄).
Non-Oxidizing: Unlike sulfuric or nitric acid, it does not cause unwanted side reactions.
High Thermal Stability: Stable up to ~300°C.
Good Solubility: Miscible with water and polar organic solvents (acetonitrile, DCM, DMF).
Low Nucleophilicity: The conjugate base (CF₃SO₃⁻, triflate) is weakly coordinating, ideal for carbocation-based reactions.3. Key Specifications with Explanation
| Parameter | Typical Value | Explanation |
|---|---|---|
| Appearance | Colorless to pale yellow liquid | Darkening suggests decomposition or impurities. |
| Assay (Purity) | ≥98.0% – ≥99.5% | High purity essential for sensitive synthesis and electronics. |
| Molecular Weight | 150.08 g/mol | Required for molar calculations. |
| Boiling Point | 162°C (at 760 mmHg) | Distillable under normal pressure. |
| Melting Point | -40°C | Remains liquid at low temperatures. |
| Water Content (KF) | ≤0.1% – ≤0.05% | Critical for moisture-sensitive reactions (electronics/OLED). |
| Sulfate (SO₄²⁻) | ≤0.05% | High sulfate can poison catalysts in downstream reactions. |
| Density | 1.696 g/mL (20°C) | Heavier than water. |
| pKa | ≈ -14 | Extreme acidity; protonates nearly all organic functional groups. |
4. Applications
| Application Area | Specific Uses |
|---|---|
| Organic Synthesis | Protonation of inert substrates (alkanes, ketones); Friedel-Crafts acylation/alkylation; Beckmann rearrangement. |
| Polymer Chemistry | Initiator for cationic polymerization (e.g., polyisobutylene, polyoxymethylene). |
| Pharmaceuticals | Protecting group chemistry; synthesis of APIs (active ingredients). |
| Battery Electrolytes | Component in lithium-ion batteries (LiTFSI) and supercapacitors. |
| Electronics | Photoresist formulation for semiconductor manufacturing (OLED, IC). |
| Petrochemicals | Alkylation catalyst for high-octane gasoline production. |
5. Grade Comparison
| Grade | Purity | Water Content | Typical Use |
|---|---|---|---|
| Industrial Grade | ≥98.0% | ≤0.2% | Petrochemical alkylation, bulk synthesis. |
| Laboratory / Reagent Grade | ≥99.0% | ≤0.1% | General R&D, pharmaceutical intermediates. |
| High Purity / Electronic Grade | ≥99.5% | ≤0.02% | OLED, IC photoresists, Li-battery electrolytes. |
6. Buying Guide
Select Grade by Application:
Bulk alkylation / industrial catalysis → Industrial grade (≥98%).
Pharma / fine chemical synthesis → Reagent grade (≥99%, low sulfate).
Electronics / battery applications → Electronic grade (≥99.5%, <0.02% H₂O).
Packaging & Handling:
Highly corrosive – requires PTFE or glass-lined containers.
Avoid metal contact – will corrode stainless steel (use Hastelloy, PTFE, or glass).
Fuming liquid – handle under inert atmosphere (N₂/Ar) in a fume hood.
Storage Conditions:
Store in PTFE, glass, or Hastelloy containers (no metal caps).
Keep tightly sealed, away from moisture and bases.
Ambient temperature is acceptable; refrigeration not required.
Safety Verification:
Request CoA confirming purity, water content, and sulfate level.
Ensure supplier provides detailed MSDS with corrosion handling instructions.
7. FAQ
Q: Why is triflic acid stronger than sulfuric acid?
A: The triflate ion (CF₃SO₃⁻) is extremely stable due to the strong electron-withdrawing effect of the -CF₃ group and resonance delocalization. This stabilizes the conjugate base, making the acid much stronger.
Q: What materials are compatible with triflic acid?
A: Compatible: PTFE (Teflon), PFA, glass (borosilicate), glass-lined steel, Hastelloy C-276, tantalum.
Not compatible: Carbon steel, stainless steel (304/316), aluminum, most plastics (PE, PP, PVC).
Q: Is triflic acid dangerous?
A: Yes, highly corrosive. GHS: H290 (Corrosive to metals), H302 (Harmful if swallowed), H314 (Severe skin burns/eye damage). Always use full PPE (acid-resistant gloves, face shield, lab coat) in a fume hood.
Q: How is triflic acid different from methanesulfonic acid (MSA)?
A: Triflic acid (CF₃SO₃H) is ~1000x stronger than MSA (CH₃SO₃H) due to fluorine substitution. MSA is much weaker (pKa ≈ -2) and less corrosive.
Q: Can I dilute triflic acid with water?
A: Yes, but always add acid to water slowly with cooling – extreme heat is generated (exothermic). Never add water to concentrated acid. Diluted solutions remain strongly acidic.
Q: How should I dispose of triflic acid?
A: Neutralize carefully with sodium bicarbonate or sodium hydroxide (under fume hood, with cooling), then dispose as corrosive aqueous waste. Never pour concentrated acid down the drain.
8. Delivery, Certification & Service
Packaging: 100g, 500g, 1kg PTFE-coated bottles; 25kg, 200kg drum (Hastelloy/PTFE lined).
Lead Time: Stock items: 3–5 business days; custom packaging: 2–3 weeks.
Documentation: CoA (purity, water content, sulfate), MSDS, TDS.
Certifications: ISO 9001; Electronic grade meets SEMI standards.
Restricted Shipping: Class 8 (Corrosive) – UN 3265. Requires hazardous material documentation.