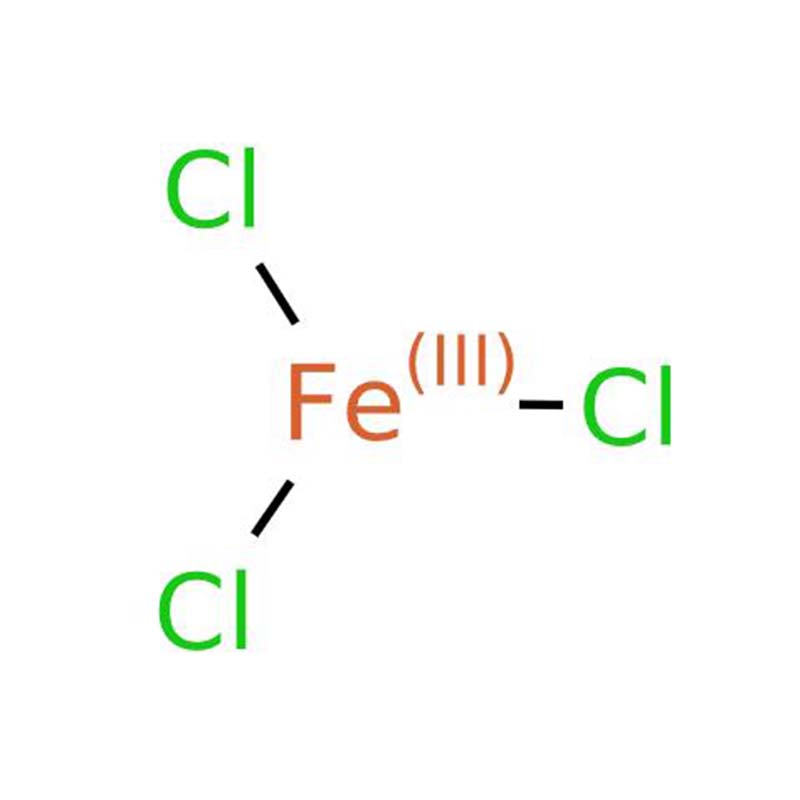
Ferric chloride
High-Efficiency Water Treatment: Outperforms alum in removing suspended solids, organic pollutants, and phosphates, drastically reducing water turbidity.
Versatile Industrial Use: Acts as a powerful catalyst and oxidizer for organic synthesis and dye production, with broad solvent compatibility.
Novel Antibacterial Application: Triggers ferroptosis to combat drug-resistant pathogens (e.g., Pseudomonas aeruginosa), offering new options for wound infection treatment.
Bulk & Cost-Effective Supply: Provided in FCL quantities to support large-scale industrial operations at competitive costs.
Ferric chloride appears as a dark brown, hexagonally structured crystalline solid. It shows a deep reddish-purple tint when light passes through it and exhibits a metallic green luster under direct light irradiation. This substance dissolves readily in water, methanol, ethanol, acetone and diethyl ether; it is also soluble in liquid sulfur dioxide, phosphorus tribromide, phosphorus oxychloride, ethylamine and aniline. It is sparingly soluble in carbon disulfide and insoluble in glycerol. Its main applications cover water treatment, and it also functions as a mordant, catalyst, chlorinating reagent as well as a raw material for preparing other iron-containing salts.
| Melting point | 304 °C(lit.) |
| Boiling point | 316 °C |
| density | 2,804 g/cm3 |
| bulk density | 1000kg/m3 |
| vapor density | 5.61 (vs air) |
| vapor pressure | 1 mm Hg ( 194 °C) |
| refractive index | n20/D1.414 |
| Fp | 316°C |
| storage temp. | Store below +30°C. |
| solubility | H2O: soluble |
| form | powder |
| color | Yello |
| w | |
| Specific Gravity | 2.804 |
| PH | 1 (200g/l, H2O, 20℃) |
| Water Solubility | 920 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| Crystal Structure | BiI3 type |
| crystal system | Three sides |
| Merck | 144,019 |
| Space group | R3 |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm30.6060.6061.74190901200.5529 |
| Exposure limits | ACGIH: TWA 1 mg/m3 |
| NIOSH: TWA 1 mg/m3 | |
| Stability: | Stable. Very sensitive to moisture. Incompatible with strong oxidizing agents; |
| forms explosive mixtures with sodium, potassium. Hygroscopic. | |
| CAS DataBase Reference | 7705-08-0(CAS DataBase Reference) |
| NIST Chemistry Reference | Ferric chloride(7705-08-0) |
| EPA Substance Registry System | Ferric chloride (7705-08-0) |
Safety Information
| Hazard Codes | C,Xn,Xi |
| Risk Statements | 41-38-22-34-37/38-10-36 |
| Safety Statements | 26-39-45-36/37/39 |
| RIDADR | UN 2582 8/PG 3 |
| WGK Germany | 1 |
| RTECS | LJ9100000 |
| TSCA | Yes |
| HS Code | 2827 39 20 |
| HazardClass | 8 |
| PackingGroup | III |
| Hazardous Substances Data | 7705-08-0(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 316 mg/kg |