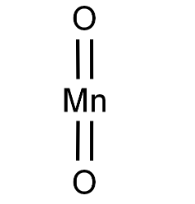Manganese dioxide CAS#1313-13-9
CAS Number: 1313-13-9
Chemical Formula: MnO2
Synonyms:
Manganese(IV) oxide, powder, 80-85%
Manganese(IV) oxide, -100 mesh, 99+%
Manganese(IV) oxide, powder, 80-85%
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Gray Powder
Manganese dioxide CAS#1313-13-9
Manganese dioxide, mainly used as a depolarizer in dry cell batteries, is the most important non-metallic form of manganese. In either traditional alkaline and rechargeable battery cells, Manganese dioxide takes in electrons through a redox reaction, becoming Manganese III oxide. It also has applications in lithium-ion technologies.
It is also the primary precursor to ferromanganese, an alloy of iron used for its lower melting point and low cost.
It is black in appearance and used to color glass and ceramics, as well as for cave art and possibly body paint in prehistorical eras. Further uses include the purification of drinking water, and production of pure oxygen and chlorine.
Manganese dioxide is a black crystalline solid. Molecular weight= 86.94; Freezing/Melting point=(decomposes)=53℃. Hazard Identification (based on NFPA-704 M Rating System): Health 3, Flammability 0, Reactivity×(Oxidizer). Insoluble in water.
Application of Manganese dioxide
Manganese(IV) oxide is the most important ore of manganese from which the metal is mostly manufactured. The oxide occurs in nature as the mineral pyrolusite as heavy gray lumps, or black when powdered.
The mineral is used to produce manganese metal, most manganese salts, and also manganese steel and other alloys. The metallurgical applications of manganese(IV) oxide mainly involve making ferromanganese and special manganese alloys. Another important application of manganese(IV) oxide is in manufacturing dry-cell batteries and alkaline cells. The oxide also is a colorant in brick, tile, porcelain and glass; a drier for paints and varnishes; a 552 MANGANESE(IV) OXIDEpreparation for printing and dyeing textiles; a curing agent for polysulfide rubbers; an adsorbent for hydrogen sulfide and sulfur dioxide; an oxidizing agent in many organic syntheses such as quinone and hydroquinone; and a catalyst in laboratory preparation of oxygen from potassium chlorate. Manganese(IV) oxide also is used to make welding rods and fluxes, and ceramic magnets (ferrites); and is an additive to fertilizers.
Manganese dioxide (MnO2) is soluble in water and HNO3 and soluble in HCl. It occurs in nature as the blue-black mineral pyrolusite. In glass, manganese dioxide is used as a colorant and decolorizer.
The major use of manganese oxides is an ore of manganese for the manufacturing of steel; manganese serves to increase the hardness and decrease the brittleness of steel. Another important use of manganese oxides is as the cathode material of common zinc/carbon and alkaline batteries (such as flashlight batteries).
As an oxidizing agent, source of metallic manganese, decolorizing glass, painting porcelain, as an analytical reagentManganese(IV) oxide is used as an oxidizing agent in organic synthesis such as oxidation of allylic/benzylic alcohols, as a textile dye, as a reducing agent, and as a component of dry cell batteries such as zinc-carbon battery and alkaline battery. It is also used in making pigments for glasses and ceramics, and drier for paints. Further it is used in the manufacture of manganese steel and several manganese derivatives including potassium permanganate, a powerful versatile oxidant. Catalytic hydration of nitriles to amides by flowing through manganese dioxide has been reported. Manganese dioxide supported on inorganic oxide can be used for oxidation of methylamine through CWAO (Catalytic Wet Air Oxidation). It has high potential as highly efficient and robust material for water oxidation reaction (WORs).
| Manganese dioxide Chemical Properties |
| Melting point | 535 °C (dec.) (lit.) |
| bulk density | 600-800kg/m3 |
| density | 5.02 |
| vapor pressure | 0-0Pa at 25℃ |
| storage temp. | Store below +30°C. |
| solubility | <0.001g/l insoluble |
| form | powder |
| color | gray |
| Specific Gravity | 5.026 |
| Water Solubility | insoluble |
| Crystal Structure | Rutile type |
| crystal system | square |
| Merck | 14,5730 |
| Space group | P42/mnm |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm30.43980.43980.28739090900.0556 |
| Exposure limits | ACGIH: TWA 0.02 mg/m3; TWA 0.1 mg/m3 OSHA: Ceiling 5 mg/m3 NIOSH: IDLH 500 mg/m3; TWA 1 mg/m3; STEL 3 mg/m3 |
| Stability: | Stable. Incompatible with strong acids, strong reducing agents, organic materials. |
| Cosmetics Ingredients Functions | ANTIOXIDANT |
| InChI | 1S/Mn.2O |
| InChIKey | NUJOXMJBOLGQSY-UHFFFAOYSA-N |
| SMILES | O=[Mn]=O |
| CAS DataBase Reference | 1313-13-9(CAS DataBase Reference) |
| NIST Chemistry Reference | manganese(IV) dioxide(1313-13-9) |
| EPA Substance Registry System | Manganese dioxide (1313-13-9) |
| Safety Information |
| Hazard Codes | Xn |
| Risk Statements | 20/22 |
| Safety Statements | 25 |
| RIDADR | 3137 |
| WGK Germany | 1 |
| RTECS | OP0350000 |
| TSCA | TSCA listed |
| HS Code | 2820 10 00 |
| PackingGroup | III |
| Storage Class | 13 - Non Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Inhalation Acute Tox. 4 Oral STOT RE 2 Inhalation |
| Toxicity | LD50 orally in rats: >40 mmole/kg (Holbrook) |
Fact Factory and Equipment Show