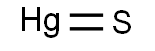Mercury(II) sulfide CAS#1344-48-5
CAS Number: 1344-48-5
Chemical Formula: HgS
Synonyms:
Mercurysulfide
mercurysulfide(hgs)
mercurysulphide
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Red Powder
Mercury(II) sulfide CAS#1344-48-5
Mercury(II) sulfide has several modifications, the only two stable allotropic forms are:
1. the red hexagonal form known as cinnabar (alpha form), and 2. the black cubic modification (beta form).
Cinnabar is a red crystalline or powdery substance; hexagonal crystal system; refractive index 2.854; density 8.10 g/cm3; sublimes at 583.5°C; color changes to brown at 250°C and converts to black sulfide at 386°C; reverts to red color on cooling; insoluble in water, alcohol and nitric acid; soluble in aqua regia and solutions of alkali metal sulfides; decomposed by hot concentrated sulfuric acid.
Black sulfide is a black amorphous powder or crystalline substance (beta form); cubic structure; metastable at ordinary temperatures; converts to red sulfide by sublimation at ordinary pressure; density 7.73 g/cm3; melts at 583.5°C; insoluble in water, alcohol and nitric acid; soluble in aqua regia, alkalies, and solutions of alkali metal sulfides.
Red sulfide occurs natively and is mined from mineral cinnabar. Also it can be prepared by heating mercury with a solution of potassium pentasulfide, producing a scarlet cinnabar:
Hg + K2S5 → HgS + K2S4
The red sulfide also may be made from black sulfide by heating in a concentrated solution of alkaline polysulfide. The shade of pigment varies with temperature, reaction time, and concentration of the black sulfide.
Alternatively, vermilion may be made by grinding sodium sulfide with sulfur and slowly adding mercury. The shades are not bright when prepared at 0°C.
The black mercury(II) sulfide is prepared usually by precipitation from an aqueous solution of mercury(II) salt with hydrogen sulfide. Thus, when H2S is passed into a solution of HgCl2, a pale yellow precipitate of composition HgCl2•2HgS forms. This converts to amorphous black HgS on further treatment with H2S.
The black sulfide may also be made by other methods such as adding sodium thiosulfate, Na2S2O3 in excess to a dilute solution of sodium mercurichloride, Na2HgCl4 and treating mercury with molten or powdered sulfur.
Application of Mercury(II) sulfide
Mercuric sulfide (HgS) is a fine, very brilliant scarlet powder that is deadly if ingested. Also known as the mercury ore cinnabar and metacinnabar, it is used as a pigment in the manufacture of paints.
Mercury(II) sulfide is used in paint, sealing wax and preservatives.
For coloring plastics, sealing wax, and with FeSO4 for marking linen; manufacture of fancy colored papers; as pigment.
| Mercury(II) sulfide Chemical Properties |
| Melting point | 583.5 °C(lit.) |
| Boiling point | 584°C |
| density | 8.1 g/mL at 25 °C |
| solubility | insoluble in H2O; soluble in acid solutions, ethanol |
| form | Powder |
| color | red |
| Water Solubility | Soluble in water (0.00001 g/L). |
| Crystal Structure | Cubic, Sphalerite Structure - Space Group F(-4)3m |
| crystal system | Three sides |
| Merck | 14,5888 |
| Space group | P3121 |
| Solubility Product Constant (Ksp) | pKsp: 52.4 |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm30.413170.413170.9445190901200.1396 |
| Exposure limits | ACGIH: TWA 0.025 mg/m3 (Skin) NIOSH: IDLH 10 mg/m3; TWA 0.05 mg/m3; Ceiling 0.1 mg/m3 |
| Stability: | Stable. |
| InChI | 1S/Hg.S |
| InChIKey | QXKXDIKCIPXUPL-UHFFFAOYSA-N |
| SMILES | S=[Hg] |
| Hardness, Mohs | 3.0 |
| CAS DataBase Reference | 1344-48-5(CAS DataBase Reference) |
| EPA Substance Registry System | Mercury sulfide (HgS) (1344-48-5) |
| Safety Information |
| Hazard Codes | N-T,Xi |
| Risk Statements | 50/53-36-33-23/24/25-43-31 |
| Safety Statements | 61-60-45-36/37/39-28A-26-36/37 |
| RIDADR | 2025 |
| WGK Germany | - |
| RTECS | OX0720000 |
| F | 8 |
| TSCA | TSCA listed |
| HazardClass | 6.1 |
| PackingGroup | II |
| HS Code | 28521000 |
| Storage Class | 13 - Non Combustible Solids |
| Hazard Classifications | Skin Sens. 1 |
Fact Factory and Equipment Show