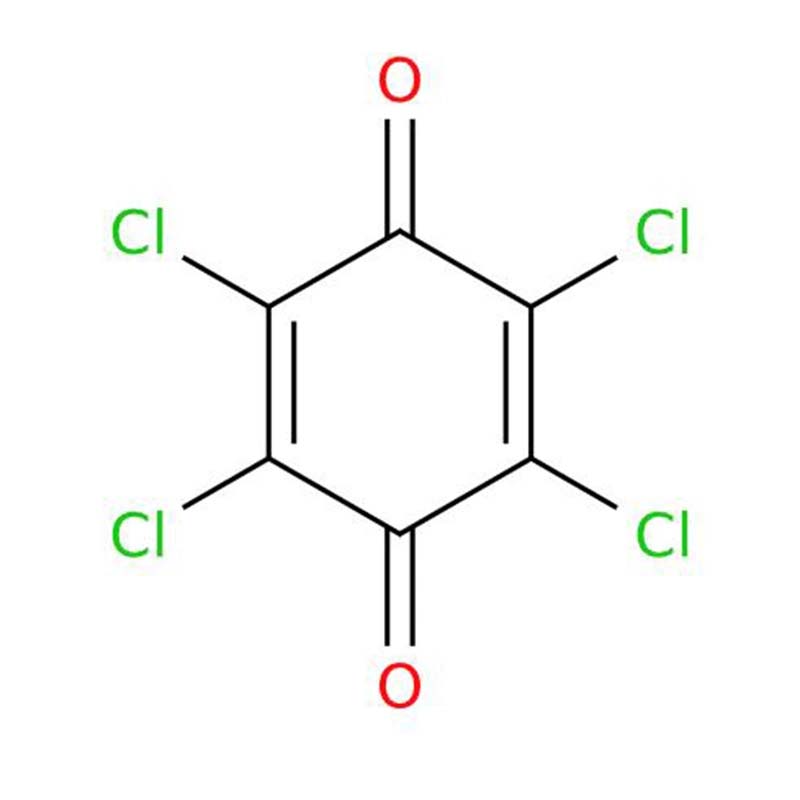
Chloranil CAS:118-75-2
Benzyl alcohol shows moderate solubility in water but mixes readily with organic solvents like ethanol, diethyl ether, benzene and chloroform, making it highly compatible with organic-phase systems.
This compound exists naturally in essential oils such as jasmine and ylang-ylang, acting as a valuable plant-sourced ingredient for perfumes and aromatic products.
With a mild and elegant floral fragrance, it is widely used in cosmetic preparations, high-end fragrances and other items demanding a gentle and refined aroma.
It has a proven safety record when used within recommended limits, yet proper handling is still recommended in industrial and cosmetic uses to avoid possible irritation to skin and mucous membranes.
Benzyl alcohol, also known as phenylmethanol with the molecular formula C₆H₅CH₂OH, has a density of 1.045 g/mL at 25°C. As a basic aromatic alcohol, its structure can be regarded as a benzene ring linked to a hydroxymethyl group, or a methanol molecule substituted by a phenyl group.
It appears as a colorless, viscous liquid with a soft aromatic scent. When stored for a long time, it may oxidize slightly to produce a small amount of benzaldehyde, which gives a typical bitter almond fragrance. This compound is polar in nature, has low toxicity and low vapor pressure, making it suitable for use as an alcohol solvent, though it is still flammable.
Its water solubility is relatively low, about 1 gram per 25 milliliters of water, while it dissolves completely in organic solvents such as ethanol, diethyl ether, benzene and chloroform. Benzyl alcohol exists naturally in many essential oils, either in free state or in ester form, including jasmine, ylang-ylang, plumeria, hyacinth, moonflower, Peru balsam and Tolu balsam.
Slow oxidation in air will produce benzaldehyde and anisole analogs, which is why many commercial products show an almond-like odor. In addition, under the action of strong oxidants such as concentrated nitric acid, benzyl alcohol can be further oxidized to benzoic acid.
Parameters
| Melting point | -15 °C |
| Boiling point | 205 °C |
| density | 1.045 g/mL at 25 °C(lit.) |
| vapor density | 3.7 (vs air) |
| vapor pressure | 13.3 mm Hg ( 100 °C) |
| FEMA | 2137 | BENZYL ALCOHOL |
| refractive index | n20/D 1.539(lit.) |
| Fp | 201 °F |
| storage temp. | Store at +2°C to +25°C. |
| solubility | H2O: 33 mg/mL, clear, colorless |
| pka | 14.36±0.10(Predicted) |
| form | Liquid |
| color | APHA: ≤20 |
| Odor | Mild, pleasant. |
| Relative polarity | 0.608 |
| explosive limit | 1.3-13%(V) |
| Odor Type | floral |
| biological source | synthetic |
| Water Solubility | 4.29 g/100 mL (20 ºC) |
| Merck | 141,124 |
| JECFA Number | 25 |
| BRN | 878307 |
| Henry's Law Constant | <2.70 x 10-7 at 25 °C (thermodynamic method-GC/UV, Altschuh et al., 1999) |
| Exposure limits | No exposure limit is set. Because of its low vapor pressure and low toxicity, the health hazard to humans from occupational exposure should be very low. |
| Dielectric constant | 13.1(20℃) |
| InChIKey | WVDDGKGOMKODPV-UHFFFAOYSA-N |
| LogP | 1.05 at 20℃ |
| Surface tension | 39.2mN/m at 293.15K |
| CAS DataBase Reference | 100-51-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Benzyl alcohol(100-51-6) |
| EPA Substance Registry System | Benzyl alcohol (100-51-6) |
Safety Information
| Hazard Codes | Xn,T |
| Risk Statements | 20/22-63-43-36/37/38-23/24/25-45-40 |
| Safety Statements | 26-36/37-24/25-23-53 |
| RIDADR | UN 1593 6.1/PG 3 |
| WGK Germany | 1 |
| RTECS | DN3150000 |
| F | 8-10-23-35 |
| Autoignition Temperature | 817 °F |
| TSCA | Yes |
| HS Code | 29062100 |
| Hazardous Substances Data | 100-51-6(Hazardous Substances Data) |
| Toxicity | LD50 orally in rats: 3.1 g/kg (Smyth) |