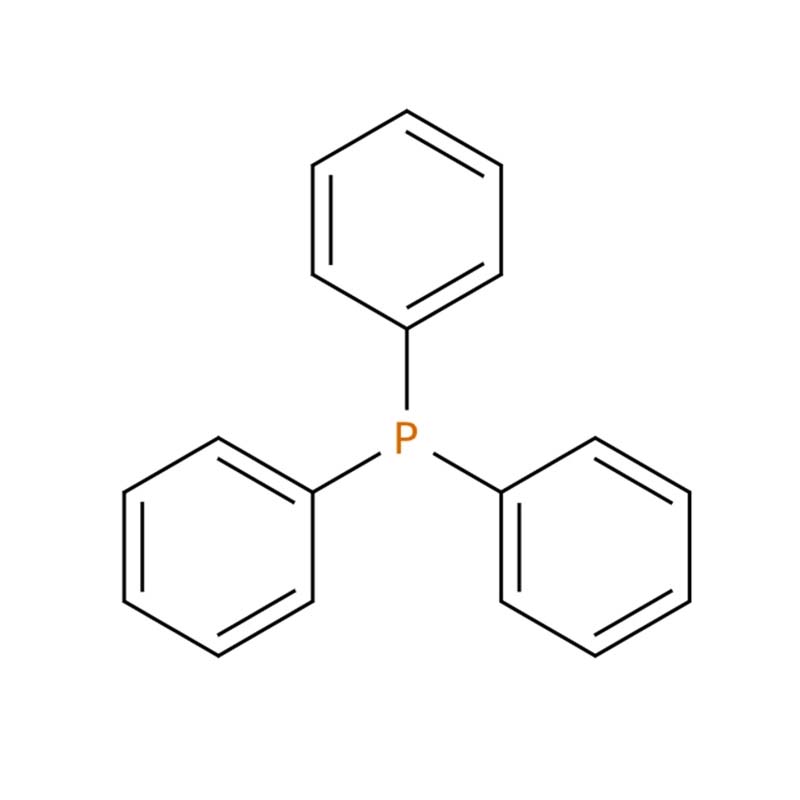
Triphenylphosphine (PPh₃) CAS: 603-35-0
Triphenylphosphine (PPh₃, CAS 603-35-0) is a white crystalline organophosphorus compound with the formula C₁₈H₁₅P . It is insoluble in water but dissolves readily in common organic solvents like ether and benzene . As a cornerstone reagent in organic and organometallic chemistry, it is valued for its strong nucleophilicity and mild reducing ability . Its primary applications include serving as an essential ligand for transition-metal catalysts (e.g., Wilkinson's catalyst and Pd-based cross-coupling systems), and as a key reactant in important name reactions like the Wittig olefination, Mitsunobu reaction, and Appel reaction .
1. Overview
Triphenylphosphine is a white crystalline solid, widely used as a ligand in transition metal catalysis and a reducing agent in organic synthesis. It is air-stable in solid form but slowly oxidizes in solution. It is a key component in the Wittig reaction, Mitsunobu reaction, and many cross-coupling processes.
2. Key Features
Excellent Ligand: Forms stable complexes with palladium, nickel, rhodium, and ruthenium for cross-coupling reactions.
Wittig Reagent Precursor: Converts to phosphonium salts for alkene synthesis from carbonyls.
Reducing Agent: Reduces organic peroxides, disulfides, and amine N-oxides.
High Purity: Available in ≥99% grades with low chloride and phosphine oxide content.
Good Solubility: Soluble in organic solvents (toluene, THF, DCM, ether); insoluble in water.
3. Key Specifications with Explanation
| Parameter | Typical Value | Explanation |
|---|---|---|
| Appearance | White crystalline powder or flakes | Yellowing indicates oxidation to triphenylphosphine oxide (TPPO). |
| Assay (Purity) | ≥99.0% | Higher purity ensures consistent catalytic activity. |
| Melting Point | 79–81°C | Narrow range indicates high purity. |
| Molecular Weight | 262.29 g/mol | Required for molar calculations. |
| Solubility | Soluble in organic solvents | Insoluble in water; facilitates easy recovery. |
| TPPO Content | ≤0.5% – ≤1.0% | Low oxide content critical for sensitive reactions. |
| Chloride (Cl⁻) | ≤0.05% | High chloride can poison metal catalysts. |
| Loss on Drying | ≤0.3% | Low moisture for moisture-sensitive applications. |
4. Applications
| Application Area | Specific Uses |
|---|---|
| Wittig Reaction | Converts carbonyls (aldehydes/ketones) to alkenes – key for vitamin A and carotenoid synthesis. |
| Mitsunobu Reaction | Couples alcohols with nucleophiles for stereochemical inversion. |
| Cross-Coupling Catalysis | Ligand for Pd-catalyzed Suzuki, Heck, Stille, and Negishi reactions. |
| Reduction Reactions | Reduces azides to amines, peroxides to alcohols, and N-oxides to amines. |
| Hydroformylation | Ligand for Rh-catalyzed hydroformylation (oxo process) in aldehyde production. |
| Polymer Chemistry | Initiator and stabilizer in various polymerization processes. |
5. Grade Comparison
| Grade | Purity | TPPO Content | Typical Use |
|---|---|---|---|
| Industrial Grade | ≥98.0% | ≤2.0% | Bulk Wittig reactions, general synthesis. |
| Reagent Grade | ≥99.0% | ≤0.5% | Cross-coupling catalysis, pharmaceutical intermediates. |
| High Purity / Electronic Grade | ≥99.5% | ≤0.1% | OLED materials, sensitive organometallic synthesis. |
6. Buying Guide
Select Grade by Application:
Bulk synthesis / Wittig reactions → Industrial grade (≥98%).
Pd-catalyzed cross-coupling → Reagent grade (≥99%, low chloride).
OLED / electronics → High purity grade (≥99.5%, very low TPPO).
Storage Conditions:
Store in cool, dry place – avoid heat and moisture.
Keep tightly sealed – protect from air (oxidizes slowly).
Under inert gas (N₂/Ar) recommended for long-term storage.
Shelf life: 12–24 months when stored properly.
Quality Checks:
Check appearance – should be white. Yellow or tan indicates oxidation to TPPO.
Request CoA verifying purity, melting point, and TPPO content.
7. FAQ
Q: How does PPh₃ differ from P(OEt)₃ or other phosphines?
A: PPh₃ is electron-donating and bulky due to three phenyl rings. P(OEt)₃ is more electron-withdrawing. Bulkier phosphines like P(t-Bu)₃ are stronger donors. PPh₃ is the most common and cost-effective for many cross-couplings.
Q: Why does my PPh₃ look yellow/sticky?
A: It has oxidized to triphenylphosphine oxide (TPPO). TPPO is less soluble, less reactive, and can poison catalysts. Discard heavily oxidized material or purify by recrystallization from hot ethanol.
Q: Is PPh₃ toxic?
A: Yes. GHS: H302 (Harmful if swallowed), H317 (May cause allergic skin reaction), H373 (May cause organ damage). Use PPE (gloves, lab coat) and work in a fume hood.
Q: Can PPh₃ be recycled?
A: Yes. TPPO (oxidation product) can be reduced back to PPh₃ using trichlorosilane (HSiCl₃) or phosgene, but this is typically done at industrial scale. Lab-scale recycling is uncommon.
Q: What solvents dissolve PPh₃?
A: Soluble: Toluene, THF, diethyl ether, DCM, ethyl acetate, acetone. Insoluble: Water, hexane (slightly).
Q: How should I store PPh₃?
A: In an amber glass or plastic bottle (light protection recommended), tightly sealed, under inert gas (N₂/Ar). Store in a cool, dry place away from oxidizing agents.
8. Delivery, Certification & Service
Packaging: 100g, 500g, 1kg, 5kg bottles; 25kg fiber drums.
Lead Time: Stock items ship within 3–5 business days.
Documentation: CoA (purity, melting point, TPPO, chloride), MSDS, TDS.
Certifications: ISO 9001 certified. Products meet ≥99% purity specification.
Shipping: Not classified as dangerous goods for most transport (check local regulations). Keep away from oxidizing agents.