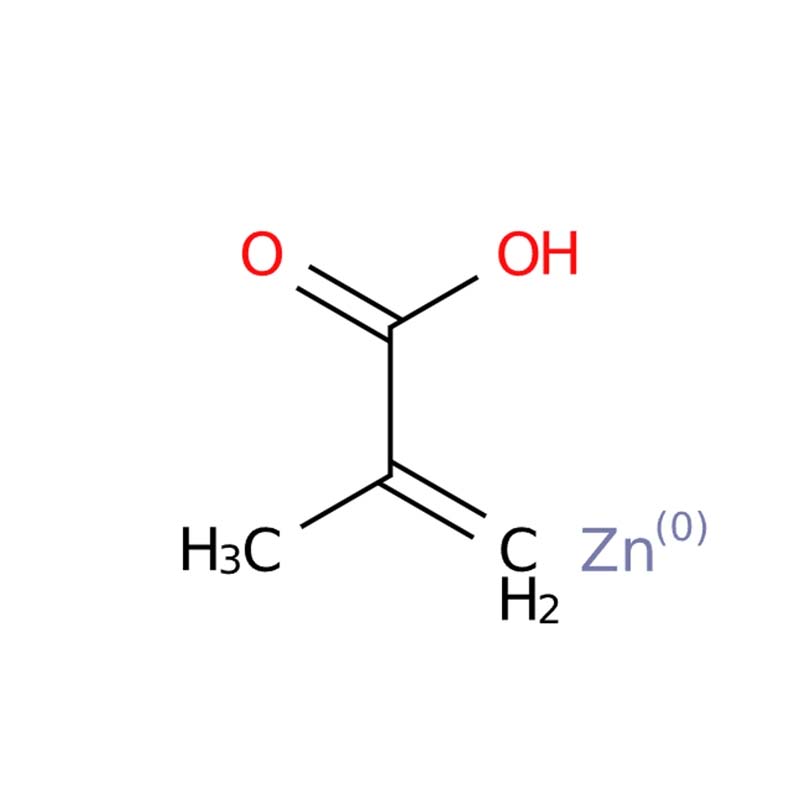
Zinc Methacrylate (ZDMA) CAS: 13189-00-9
Zinc methacrylate (ZnMA, CAS 13189-00-9) is a white to off-white crystalline powder with the formula C₈H₁₀O₄Zn . As a functional metal salt of methacrylic acid, it is valued as a highly effective co-agent and crosslinking agent in rubber and elastomer processing, where it forms ionic crosslinks that significantly improve tensile strength, tear resistance, and thermal stability of peroxide-cured vulcanizates . It also enhances adhesion to metal substrates in rubber-to-metal bonding applications . Additionally, it serves as a functional monomer in the synthesis of specialty polymers, dental resins, and ionomer-based coatings .
1. Overview
Zinc methacrylate is a reactive multifunctional salt used primarily as a scorch retarder, crosslinking agent, and coagent in rubber and polymer processing . It appears as a white powder and polymerizes upon heating or with free radical initiators. The zinc ion provides additional ionic crosslinks, enhancing mechanical properties.
2. Key Features
Dual crosslinking mechanism – covalent (via methacrylate groups) + ionic (via Zn²⁺)
Excellent scorch safety – retards premature vulcanization
Reinforcing filler – improves modulus, tensile strength, and tear resistance
Good compatibility – disperses well in diene rubbers (NR, SBR, NBR, BR)
Heat & oil resistance – enhances high-temperature performance
3. Key Specifications with Explanation
| Parameter | Typical Value | Explanation |
|---|---|---|
| Appearance | White powder | Discoloration indicates degradation or impurities. |
| Assay (Purity) | ≥95% – ≥98% | Higher purity ensures consistent crosslinking density. |
| Molecular Weight | 235.55 g/mol | Required for stoichiometric calculations. |
| Melting Point | 229–232°C (decomp.) | Decomposes before melting; indicates thermal stability . |
| Density | 1.48 g/cm³ | Affects compounding and dispersion calculations . |
| Decomposition Temp. | ≥200°C | Minimum temperature for thermal stability . |
| Zinc Content | ~27.5% (theoretical) | Active zinc available for ionic crosslinking. |
4. Applications
| Application Area | Specific Uses |
|---|---|
| Rubber Industry | Coagent for peroxide-cured elastomers (HNBR, EPDM, NBR, ACM). Improves heat resistance, modulus, and tear strength. Scorch retarder in sulfur vulcanization . |
| Adhesives & Sealants | Crosslinking agent for high-performance structural adhesives (automotive, construction) . |
| Coatings | Corrosion-resistant coatings for metal surfaces; improves adhesion and durability . |
| Dental Materials | Component in dental resins and composites; enhances mechanical properties . |
| Polymer Modification | Grafting and crosslinking agent for plastics and elastomers; improves thermal stability . |
| Synthetic Marble | Crosslinking agent for Artificial marble(artificial marble) production . |
5. Grade Comparison
| Grade | Purity | Typical Use |
|---|---|---|
| Industrial Grade | ≥95% | Rubber compounding, general crosslinking applications |
| High Purity | ≥98% | Medical devices, high-performance elastomers, dental materials |
6. Buying Guide
Select by Application:
Rubber compounding → Industrial grade (≥95%) is sufficient.
Medical/dental applications → High purity grade required; request biocompatibility data.
Peroxide-cured HNBR → Ensure low moisture and consistent particle size for dispersion.
Storage Conditions:
Store in a cool, dry place (<30°C).
Keep tightly sealed – hygroscopic and may cake.
Avoid heat, sparks, and open flames.
Shelf life: 12–24 months when stored properly.
Quality Checks:
Request CoA verifying purity, melting point, and zinc content.
Check for caking or discoloration upon receipt.
7. FAQ
Q: What is the difference between zinc methacrylate and zinc diacrylate (ZDA)?
A: Zinc methacrylate (ZDMA) has methyl groups on the double bond, providing better scorch safety and slower cure. Zinc diacrylate (ZDA) is more reactive, cures faster, and gives higher modulus but less scorch protection.
Q: How is ZDMA used in rubber compounding?
A: Typical loading: 5–30 phr. Added with peroxide curing systems. Activates at curing temperature (~150–180°C). Improves heat resistance, tensile strength, and compression set.
Q: Is ZDMA hazardous?
A: Yes. GHS: H302 (Harmful if swallowed), H317 (May cause allergic skin reaction), H318 (Causes serious eye damage), H400 (Very toxic to aquatic life) . Use PPE (gloves, goggles, dust mask). Avoid environmental release.
Q: Can ZDMA be used in food contact applications?
A: Check local regulations (FDA, EU 10/2011). Generally requires high-purity grade and specific migration testing.
Q: How does ZDMA compare to other coagents (e.g., TAC, TAIC, HVA-2)?
A: ZDMA provides ionic crosslinks in addition to covalent bonds, giving unique reinforcement. TAC/TAIC are purely covalent coagents. ZDMA is preferred for high-temperature oil-resistant applications (e.g., automotive seals).
Q: Is ZDMA soluble in water or organic solvents?
A: Slightly soluble in water; insoluble in most organic solvents. Disperses as fine particles in rubber compounds.
8. Delivery, Certification & Service
Packaging: 25kg bag (multi-wall paper with PE liner), 500kg bulk bag
Lead Time: Stock items: 3–7 business days; custom packaging: 2–3 weeks
Documentation: CoA (purity, melting point, zinc content), MSDS, TDS
Certifications: ISO 9001 (varies by supplier)
Shipping: Not classified as hazardous for most transport; check local regulations