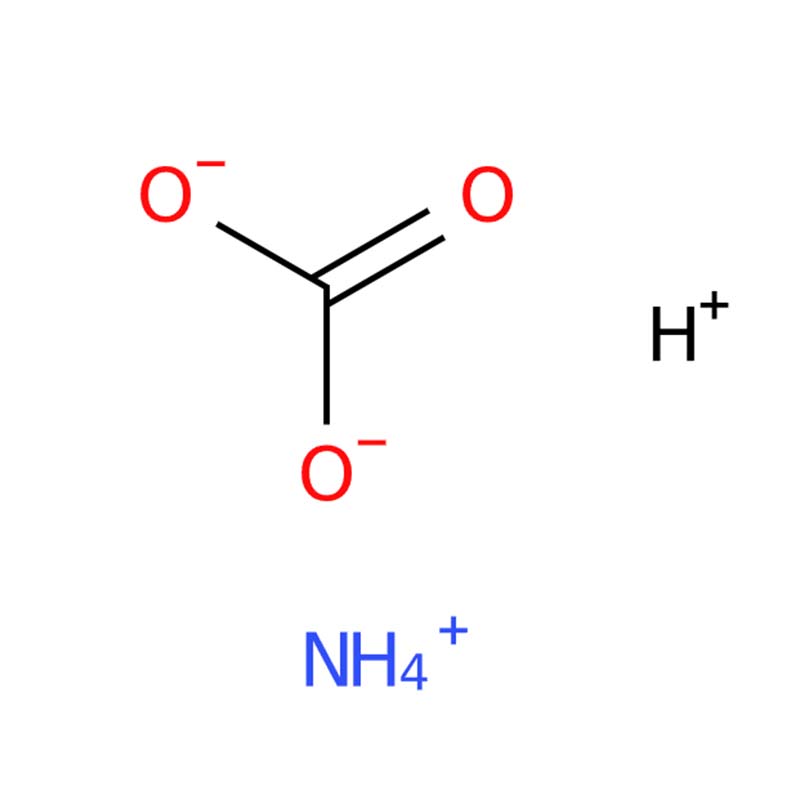
Ammonium Bicarbonate CAS : 1066-33-7
Ammonium Bicarbonate (NH₄HCO₃), commonly known as hartshorn or powdered baking ammonia, is a white crystalline compound. Its key property is thermal decomposition, rapidly releasing ammonia, carbon dioxide, and water vapor when heated. This makes it widely useful as a leavening agent in the food industry (e.g., for cookies, crackers) and as a fast-acting nitrogen fertilizer in agriculture. It is unstable at room temperature and must be stored sealed in a cool, dry place.
I. Basic Characteristics Overview
II. Key Physical & Chemical Properties
| Property | Value / Description |
| Chemical Formula | NH₄HCO₃ |
| Molar Mass | 79.06 g/mol |
| Appearance | White crystalline granules or fine powder |
| Odor | Distinct, irritating ammonia smell |
| Density | Approximately 1.586 g/cm³ (solid) |
| Water Solubility (20°C) | ~22 g/100 mL; solubility rises noticeably with higher temperature |
| pH (1% aqueous solution) | Around 7.8, showing weak alkalinity |
| Thermal Behavior | Thermally unstable; starts decomposing at ~36°C, releasing NH₃, CO₂ and H₂O vapor |
| Chemical Reactivity | Reacts readily with acids to produce CO₂ gas; reacts with strong bases to release ammonia; undergoes gradual decomposition in humid air |
III. Primary Application Fields
Food Industry (Core Application)
Commonly known as "baking ammonia", it functions as a leavening agent in baked goods like cookies and crackers. Upon heating, it decomposes into ammonia and carbon dioxide gas, which causes dough to rise quickly, forming a light, porous texture. Since all decomposition products are volatile, it is ideal for low-moisture, high-temperature baked items where a crisp texture is required.
Agriculture
Used as a fast-acting nitrogen fertilizer. It leaves no acidic residue after application and adapts well to various soil types and crops. To minimize ammonia volatilization losses, deep placement with proper soil covering is recommended.
Chemical Manufacturing
Acts as a foundational raw material for producing other ammonium salts (such as ammonium carbonate and ammonium sulfate), as well as carbonates, foamed plastics, and rubber products.
Pharmaceuticals & Laboratory Reagents
Serves as a raw material or intermediate in pharmaceutical synthesis, and also functions as an analytical reagent and pH buffer in lab applications.
Fire Protection
Incorporated as a component in certain dry powder fire extinguishing agents.
Other Industrial Uses
Applied in ceramics production, leather tanning, textile dyeing, and other processing sectors.
IV. Storage & Handling
Storage Conditions
Must be kept in a cool, dry, and well-ventilated warehouse. The ambient temperature should be controlled below 30°C, and relative humidity maintained under 75%. Protect the material from direct sunlight and heat sources at all times.
Packaging
Should be sealed in plastic-lined woven bags or airtight plastic containers to avoid moisture absorption and contact with air.
Handling Precautions
Operators must wear dust masks, chemical safety goggles, and rubber gloves to prevent inhalation of dust and skin contact.
Handle carefully during transportation to avoid damaging the packaging.
Ensure work areas are equipped with effective local exhaust ventilation to prevent buildup of ammonia vapor.
Do not store or transport together with strong acids, strong bases, or moisture-sensitive materials.
Spill Management
Use a clean shovel to collect spilled material into a dry, covered container. Rinse the spill site thoroughly with large amounts of water; the diluted wastewater can then be discharged into the industrial wastewater system as per local regulations.