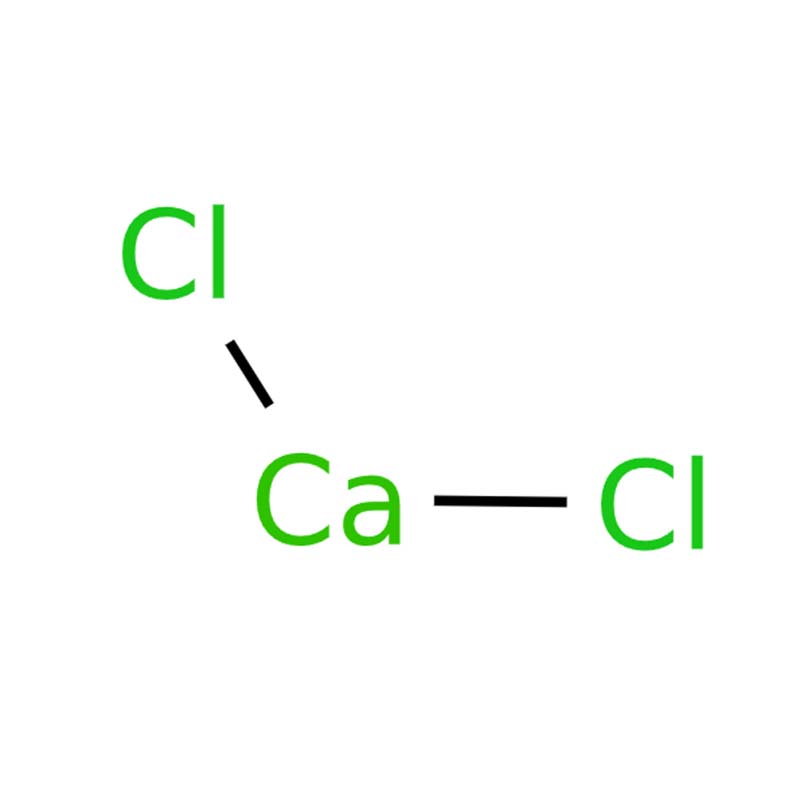
Calcium dichloride CAS: 10043-52-4
Versatile Refrigeration & Deicing Agent:Commonly used in brine systems for refrigeration equipment and as an effective road de-icing agent.
Highly Efficient Desiccant:Readily absorbs moisture from the air and deliquesces, making it ideal for drying applications (anhydrous form requires sealed storage).
Broad Food & Industrial Compatibility:Its hydrates and solutions are widely used in food manufacturing, meeting relevant quality and safety requirements.
Multi-Industry Utilit:Extends significant applications to building materials, medicine, biology, and other industrial fields.
Calcium dichloride (CaCl₂), an inorganic salt, is commonly encountered as white crystalline granules, flakes or powder. It is odorless, highly hygroscopic, and readily absorbs moisture from air to deliquesce into liquid form. The anhydrous form exhibits a rapid and strong moisture-binding capacity, requiring sealed storage to maintain stability. With a melting point of 772°C and a boiling point of 1935°C, it shows excellent thermal stability. It is freely soluble in water, releasing significant heat during dissolution, and also dissolves in ethanol and acetic acid. Its aqueous solution is neutral or slightly alkaline, with a specific gravity varying with concentration.
| Parameter | Value |
| Melting Point | 772 °C (anhydrous, pure) |
| Boiling Point | 1935 °C (anhydrous, 1 atm) |
| Density (25°C) | 2.15 g/cm³ (anhydrous); 1.086 g/mL (20°C solution) |
| Solubility in Water | 74.5 g/100 mL (20°C, highly exothermic dissolution) |
| Hygroscopicity | Strong deliquescence, absorbs >1.5× its mass of water at high humidity |
| Refractive Index (aqueous) | n₂₀/D 1.358 (standard solution) |
| Thermal Stability | Stable up to 1600°C; dihydrate fully dehydrates at ~260°C |
| pH (10% aqueous solution) | Neutral to slightly alkaline (6.8–8.0) |
- Food Industry
Processing Aid: Functions as a firming agent and preservative in canned fruits/vegetables, maintaining texture and extending shelf life at low concentrations.
Beverage & Dairy Stabilizer: Adjusts mineral balance and enhances stability in beer, cheese, and sports drinks.
Desiccant for Packaging: Controls moisture in food-grade packaging to prevent caking and spoilage of dry products.
- Refrigeration & HVAC
Brine System Fluid: Serves as a primary component in low-temperature refrigeration brines, enabling efficient heat transfer at sub-zero working temperatures.
Dehumidification Agent: Removes excess moisture in industrial air systems to prevent condensation and equipment corrosion.
- Road & Infrastructure Maintenance
De-icing & Anti-icing Agent: Melts ice and snow effectively at temperatures as low as -25°C, with less corrosive impact than traditional salts.
Dust Control: Suppresses road dust by binding fine particles, improving air quality and road safety.
- Construction & Building Materials
Concrete Accelerator: Speeds up cement hydration and early strength development, ideal for cold-weather construction projects.
Waterproofing Additive: Reduces permeability in concrete and mortar, enhancing structural durability.
- Chemical & Industrial Manufacturing
Desiccant for Gas Drying: Dehydrates natural gas, air, and other industrial gases by absorbing moisture efficiently.
Chemical Synthesis Intermediate: Used in the production of calcium salts, dyes, and specialty chemicals.
Water Treatment: Adjusts water hardness and precipitates impurities in industrial wastewater and municipal treatment systems
Calcium dichloride (CaCl₂, CAS: 10043-52-4) is commonly supplied in various forms, including white crystalline granules, flakes, or powder, with different hydration levels (anhydrous, dihydrate, hexahydrate) available for diverse applications. The anhydrous form has a high melting point of 772°C and excellent thermal stability, while its hydrates show different solubility and moisture-binding properties. The strong ionic hydration effect contributes to its high solubility and exothermic dissolution behavior, which is widely utilized in refrigeration brines, de-icing solutions, and industrial drying systems.