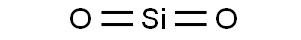Quartz CAS#14808-60-7
CAS Number: 14808-60-7
Chemical Formula: O2Si
Synonyms:
Quartz (SiO2)
Rose quartz
silica,crystalline(asrespirabledust)
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: White Powder
Quartz CAS#14808-60-7
Silicon dioxide (chemical formula: SiO2) is a chemical compound comprised of oxide and silicon elements. It is insoluble in water or acids except hydrofluoric acid. It appears as transparent to gray, tasteless crystal which is widely presented in nature as sand or quartz. It is also one of the most abundant mineral existing in Earth’s crust. It has wide applications in various fields as below: (1) as the precursor to glass and silicon; (2) for sand casting; (3) as a additive to food and pharmaceutical products; (4) for production of cement and refractory materials; (5) as the medium for conversion of energy. (6) Extraction of DNA and RNA; (7) as a defoamer component. There are many ways for production of silicon. The most common way is through mining and purification of quartz.
Quartz is a colorless solid that exists in numerous crystalline forms. Oxygen and silicon are the two most common elements in the earth's crust, and Quartz is the principal component of sand. Quartz is used biologically, most notably by phytoplankton diatoms and the zooplankton radiolarians in their shells. Quartz, SiO2, should not be confused with Quartz or silicones. Quartz contain the basic tetrahedral unit SiO44-bonded to metal ions such as aluminum, iron, sodium, magnesium, calcium, and potassium to form numerous Quartz minerals. Silicones are synthetic polymers made of monomers with at least two silicon atoms combined with an organic group and generally containing oxygen.
Application of Manganese dioxide
As the art of glass making developed, individuals discovered how to produce different glasses by adding various substances to the Quartz melt. The addition of calcium strengthened the glass, and other substances imparted color to the glass. Iron and sulfur give brown glass, copper produces a light blue color, and cobalt a dark blue color. Manganese was added to produce a transparent glass, and antimony to clear the glass of bubbles. Most modern glass produced is soda-lime glass and consists of approximately 70% SiO2, 15% Na2O (soda), and 5% CaO (lime). Borosilicate glass is produced by adding about 13% B2O3. Borosilicate glass has a low coeffi cient of thermal expansion and is therefore very heat resistant. It is used extensively in laboratory glassware and in cooking where it is sold under the brand name Pyrex. Because of Quartz's high melting point, it is ideal for making molds for metal casting. It is regularly used to form iron, aluminum, and copper items. Quartz is the primary filter medium used in wastewater treatment. Filtration systems often modify Quartz physically and chemically to produce activated Quartz formulations. Besides water treatment, activated Quartz gels used for chromatography in chemistry laboratories. In the construction industry, Quartz glass is used as fiber glass insulation, Quartz sand is a basic ingredient in cement and concrete, and is used indirectly in building products. Quartz is used as filler in paints, adhesives, rubber, and coatings. It is added to personal care products such as tooth polishes.
Electronic components; piezoelectric control in filters, oscillators, frequency standards, wave filters, radio and TV components; barrel-finishing abra- sive.
Formulators may select Quartz as an alternative to diatomaceous earth or clay, particularly when developing mineral make-up, pressed or loose powders. Quartz has abrasive, absorbent, and anti-caking properties, and can also reduce the transparency of a formulation. Quartz is the U.S. name for what is known as solum diatomeae.
| Quartz Chemical Properties |
| Melting point | 1610 °C(lit.) |
| Boiling point | 2230 °C |
| bulk density | 1400kg/m3 |
| density | 2.6 g/mL at 25 °C(lit.) |
| refractive index | n |
| storage temp. | no restrictions. |
| solubility | insoluble in H2O, acid solutions; soluble in HF |
| form | powder |
| color | white |
| Specific Gravity | 2.2-2.6 |
| PH | 5-8 (400g/l, H2O, 20℃)(slurry) |
| Water Solubility | insoluble |
| Thermal Conductivity | 6.485 W/(m·K) (crystal) |
| Crystal Structure | Quartz type |
| crystal system | Three sides |
| Space group | P3221 |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm30.491340.491340.5405290901200.113 |
| Exposure limits | ACGIH: TWA 0.025 mg/m3 OSHA: TWA 50 μg/m3 NIOSH: IDLH 50 mg/m3; TWA 0.05 mg/m3 |
| Dielectric constant | 4.2(0.0℃) |
| Stability: | Stable. |
| Major Application | battery manufacturing |
| Cosmetics Ingredients Functions | ABRASIVE |
| InChI | 1S/O2Si/c1-3-2 |
| InChIKey | VYPSYNLAJGMNEJ-UHFFFAOYSA-N |
| SMILES | O=[Si]=O |
| Hardness, Knoop | 666 - 902 |
| Hardness, Mohs | 7.0 |
| Hardness, Vickers | 1103 - 1260 |
| Drilling Hardness | 7648 |
| CAS DataBase Reference | 14808-60-7(CAS DataBase Reference) |
| IARC | 1 (Vol. Sup 7, 68, 100C) 2012 |
| NIST Chemistry Reference | Silicon oxide(14808-60-7) |
| EPA Substance Registry System | Quartz (14808-60-7) |
| Safety Information |
| Hazard Codes | Xn |
| Risk Statements | 36/37/38-48/20 |
| Safety Statements | 26-24/25-22 |
| OEB | D |
| OEL | TWA: 0.05 mg/m3 |
| WGK Germany | 3 |
| RTECS | ZG6800000 |
| TSCA | TSCA listed |
| HS Code | 25061000 |
| Storage Class | 6.1D - Non-combustible acute toxic Cat.3 toxic hazardous materials or hazardous materials causing chronic effects |
| Hazard Classifications | STOT RE 1 Inhalation |
| Hazardous Substances Data | 14808-60-7(Hazardous Substances Data) |
| Toxicity | LDLo intravenous in dog: 20mg/kg |
| IDLA | 25 mg/m3 |
Fact Factory and Equipment Show