Hydrogenated castor oilCAS:8001-78-3
Appearance and Properties: Presents as a transparent viscous liquid, with color ranging from nearly colorless to pale yellow and excellent clarity.
Sensory Characteristics: Possesses a subtle aroma, with a mild and slightly pungent flavor profile.
Solubility: Highly soluble in ethanol, and only partially miscible with aliphatic hydrocarbon solvents.
Room Temperature Stability: Maintains a stable liquid state under standard ambient storage conditions, without phase separation.
1. Product Overview
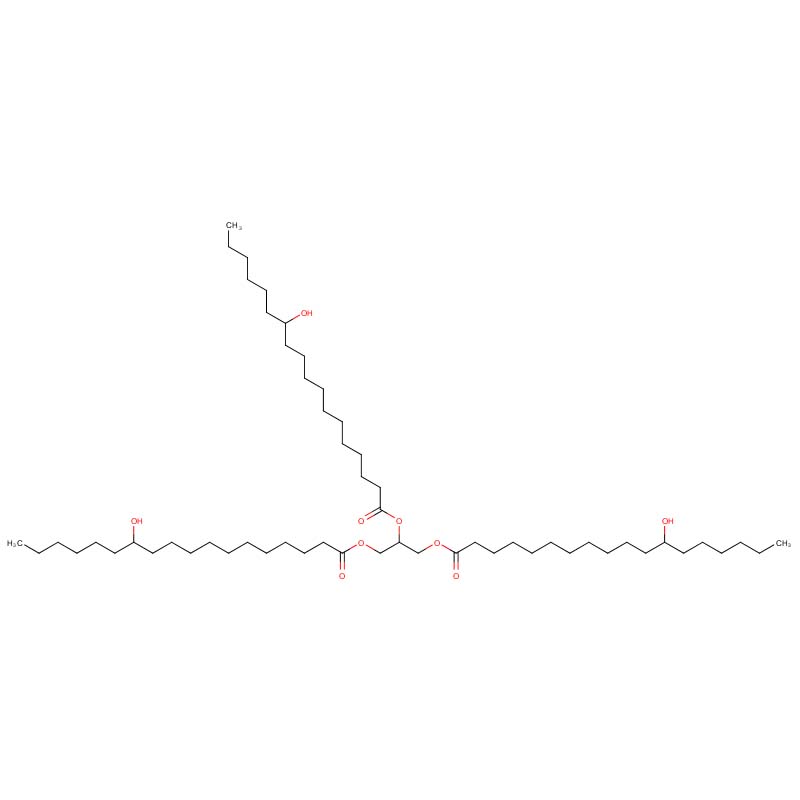
Classified as a multifunctional organic wax and rheology modifier, HCO retains the unique hydroxyl (-OH) functional groups inherent to castor oil while gaining enhanced thermal stability and a significantly elevated melting point through hydrogenation. This synergistic combination of natural origin and modified properties positions it as a staple ingredient across diverse industrial sectors, including cosmetics and personal care, pharmaceuticals, coatings and inks, plastics manufacturing, and lubricant production . As a bio-based chemical with renewable origins, HCO aligns with modern sustainability goals, serving as an indispensable natural additive in high-performance formulations due to its exceptional thickening, gelling, film-forming, and lubricating capabilities .
2. Key Features
Natural & Renewable Provenance: Derived entirely from the castor plant, HCO boasts a Renewable Carbon Index (RCI) exceeding 95% , making it a cornerstone of green chemistry initiatives and sustainable manufacturing practices .
Enhanced Melting Point & Thermal Resilience: Hydrogenation elevates its melting point to 85–88°C—markedly higher than the liquid state of unmodified castor oil—rendering it solid at ambient temperatures and ideal for heat-stable formulations .
Superior Thickening & Gelling Performance: When melted in the oil phase and cooled, HCO forms a three-dimensional network structure that substantially increases formulation viscosity and consistency. This property is critical for imparting desirable texture and long-term stability to creams, solid sticks (e.g., lipsticks, deodorant sticks), and semi-solid ointments .
Exceptional Film-Forming & Water-Resistant Traits: Upon application, HCO forms a smooth, glossy film with robust water resistance and moisturizing benefits, making it a favored component in coatings, leather care products, and skincare formulations .
High Safety & Regulatory Compliance: Approved by the FDA for use in food contact materials, HCO and its derivatives (e.g., PEGylated variants) have been deemed safe for intended use by the Cosmetic Ingredient Review (CIR) panel . It also meets the stringent standards of the United States Pharmacopeia/National Formulary (USP/NF) for pharmaceutical applications .
Multifunctional Additive Capabilities: Acts as an anti-caking agent in powders to improve flowability and release properties; functions as a lubricant and mold release agent in plastics processing; and serves as a thickener in the production of lubricating greases .
3. Technical Specifications with Explanations
Based on USP29-NF24 and relevant industrial standards, the key technical parameters of HCO are outlined below:
Parameter | Typical Value/Specification | Description & Significance |
CAS Number | 8001-78-3 (Hydrogenated Castor Oil); 8031-45-6 (sulfonated derivatives) | Universal chemical identifiers for material traceability and regulatory compliance. |
Primary Component | Glyceryl tri-12-hydroxystearate | The core functional constituent post-hydrogenation, responsible for HCO’s waxy nature and high melting point. |
Appearance | White to pale yellow powder, flakes, or waxy solid | A visual quality benchmark; different physical forms are tailored to specific processing requirements (e.g., melting vs. dry blending). |
Melting Point | 85–88°C | A defining specification: HCO remains solid below this temperature and liquefies above, critical for its role as a structuring agent and thermal stability in formulations. |
Hydroxyl Value | 154–162 mg KOH/g | A key functional metric measuring hydroxyl group content, directly influencing reactivity (e.g., with isocyanates) and compatibility in polar systems. |
Iodine Value | ≤ 5 | A primary purity indicator reflecting the degree of unsaturation; low values confirm complete hydrogenation, ensuring chemical stability and resistance to oxidation. |
Saponification Value | 176–182 mg KOH/g | Indicates the average molecular weight of triglycerides, used to verify product purity and batch consistency. |
Free Fatty Acids | ≤ 1.1 mL (0.1 N NaOH/20g) | Controls residual free fatty acid content; low levels prevent rancidity, ensuring product stability and odor neutrality. |
Density | ~0.99 g/cm³ (25°C) | Slightly less dense than water, used for volume-to-weight conversions in formulation scaling. |
Particle Size (D50) | 8.0–12.0 μm (micronized grade) | Particle size distribution for micronized variants, impacting dispersion in coatings/powders and final tactile performance. |
Biodegradability | Complies with OECD 301 standards | Demonstrates ready biodegradability under specified conditions, ensuring environmental compatibility. |
4. Applications
Cosmetics & Personal Care
Color Cosmetics: Acts as a core structuring agent and thickener in lipsticks, lip balms, foundation sticks, eye shadow sticks, and mascaras, enhancing moldability, thermal stability, and smooth application while ensuring uniform pigment dispersion .
Skin Care Products: Functions as a viscosity modifier, film former, and emollient in creams, sunscreens, and body lotions, improving texture, delivering a silky skin feel, and forming a protective barrier that boosts water resistance .
Hair Care: Provides hold and luster in styling products such as hair waxes and pomades.
Other Uses: Serves as a pigment dispersant, ensuring even distribution of colorants in the oil phase of formulations.
Pharmaceutical & Veterinary
Pharmaceutical Excipient: Utilized as a tablet lubricant and sustained-release matrix material in oral solid dosage forms. Its high melting point enables integration into hot-melt extrusion processes .
Long-Acting Injectables: In veterinary medicine, combined with hydrophobic carriers to develop long-acting injectable formulations, extending drug release for prolonged therapeutic effects .
Ointment Base: Acts as an oil phase component in semi-solid pharmaceutical preparations, supporting formulation stability and drug delivery.
Plastics & Polymer Industry
Plastic Lubricant & Mold Release Agent: In the processing of PVC, polyolefins, and other polymers, acts as both internal and external lubricant, enhancing melt flow, preventing melt fracture, and facilitating easy mold release .
Bioplastics: Serves as a processing aid and modifier for bio-based plastics, improving processability and flexibility.
Coatings & Inks
Coating Additive: In water-based and solvent-based coatings, functions as a rheology modifier, anti-settling agent, and matting agent, enhancing storage stability and application performance .
Printing Inks: Acts as a thickener and gelling agent, regulating rheology and thixotropy to improve print clarity and consistency.
Powder Coatings: Serves as a flow aid and anti-caking agent, preventing particle agglomeration and boosting spray efficiency.
Lubricating Greases & Industrial Oils
Grease Thickener: Reacts with metal hydroxides (e.g., lithium, calcium) to form 12-hydroxystearate soaps—the primary raw material for high-performance general-purpose greases (e.g., lithium-based greases) .
Metalworking Fluids: Used as a lubricity and anti-corrosion additive, enhancing tool life and workpiece quality.
Agriculture & Fertilizers
Fertilizer Anti-Caking Agent: Acts as a coating or anti-caking agent for granular fertilizers, preventing moisture absorption and caking during storage and transportation, and maintaining granule flowability .
Seed Coating: Serves as a binder and controlled-release agent in seed treatment formulations, promoting adhesion of active ingredients to seeds while providing moderate water resistance.
Food & Food Contact Applications
Food Contact Materials: FDA-approved for use in paper, paperboard, and polymer materials intended for food contact, either as an additive or coating. Its sulfonated derivatives are also utilized in food contact paper manufacturing .
Food Additive: Permitted for use as an anti-caking or surface treatment agent in select regions, subject to local regulatory requirements.
5. Product Grade Comparison
Parameter/Grade | Industrial Grade (Flakes/Solid) | Micronized Grade (Powder) | Pharmaceutical Grade (USP/NF) | Cosmetic Grade |
Core Standard | General industrial specifications | Specific particle size requirements | Meets USP/NF pharmacopeial standards | Complies with cosmetic ingredient specifications |
Physical Form | Flakes, blocks, granules | White fine powder | Flakes or powder | Flakes, powder, or granules |
Melting Point | 85–88°C | 85–88°C | 85–88°C | 85–88°C |
Particle Size (D50) | Not applicable | 8.0–12.0 μm (max ) | Not applicable | Customizable based on formulation needs |
Critical Parameters | Melting point, acid value, iodine value, saponification value | Particle size distribution, bulk density, flowability | Heavy metals, microbial limits, purity | Color, odor, heavy metal content |
Primary Applications | Grease manufacturing, plastics processing, rubber, industrial coatings | Powder coatings, fertilizer anti-caking, seed coating, premium inks | Oral solid dosage forms, long-acting injections, pharmaceutical excipients | Lipsticks, foundations, skincare products, hair waxes |
Typical Suppliers | Acme Hardesty Co., et al. | Micro Powders (NATUREFINE H325), et al. | Merck, BASF, et al. | Croda, Ashland, et al. |
Certifications/Sustainability | Standard industrial certifications | Renewable Carbon Index >95%, biodegradable certification | USP/NF compliance, GMP certification | Sustainable castor oil certifications, natural/organic compliance |
6. Purchasing & Selection Guide
Select by Physical Form
Flakes/Blocks/Solid:
Advantages: Cost-effective, convenient for packaging and transportation, ideal for large-scale melting processes.
Disadvantages: Challenging to disperse uniformly when added directly to powder mixtures.
Suitable for: Grease production, plastics processing, formulations requiring oil-phase heating.
Micronized Powder:
Advantages: Fine particle size (5–15 μm) enables room-temperature dry blending with other powders, ensuring excellent dispersion, smooth tactile feel, and glossy finish .
Disadvantages: Higher processing costs; increased surface area increases moisture absorption risk.
Suitable for: Powder coatings, fertilizer anti-caking, seed coating, premium cosmetic powders, dry-blend formulations.
Select by Purity & Application Standard
Industrial Grade: A cost-effective option for greases, plastics, and industrial coatings where strict purity requirements are not mandatory.
Cosmetic Grade: Mandatory for cosmetic formulations. Prioritize whiteness, odor (odorless or faint characteristic scent), and heavy metal content to ensure compatibility with sensory quality and safety standards .
Pharmaceutical Grade (USP/NF): Essential for oral solid dosage forms and sustained-release materials. Must meet pharmacopeial standards—request Drug Master File (DMF) and current Good Manufacturing Practices (cGMP) compliance documentation from suppliers .
Key Performance Indicator Verification
Melting Point (85–88°C): Ensures structuring capability and thermal stability in end products.
Hydroxyl Value (154–162): Critical for chemical reactions (e.g., lithium grease synthesis, polyurethane production) as it dictates formulation stoichiometry.
Iodine Value (≤5): A key marker of oxidative stability and hydrogenation completeness—lower values indicate superior stability and aging resistance.
Particle Size Distribution: For micronized grades, verify that D50 and maximum particle size align with application requirements (e.g., spray coating processes).
7. Frequently Asked Questions (FAQ)
Oil Phase Addition: Combine with oil-phase ingredients and heat to 90–95°C until fully melted and dissolved, then proceed with emulsification or mixing.
Note: HCO cannot be dissolved in cold water or room-temperature oils, as it will remain as solid particles and fail to deliver thickening or structuring effects.
A: Yes. HCO is 100% plant-derived, making it suitable for vegetarian and vegan formulations. Many suppliers offer sustainably certified castor oil-based HCO that meets natural and organic cosmetic standards .
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Regular flakes and micronized grades are available from regional warehouses for timely fulfillment.
Sample Program: Small quantities (e.g., 500g) are provided for formulation testing and performance evaluation.
Packaging Options:
Industrial grade: 20 kg multi-ply paper/woven bags, 25 kg fiber drums.
Micronized grade: 15 kg or 20 kg fiber drums or paper bags with polyethylene liners.
Custom packaging: Available for bulk orders.
Shipping: Classified as a non-hazardous material; standard freight options include express, less-than-truckload (LTL), and full truckload shipping.
8. Delivery, Certification & Service
Delivery Capabilities
Inventory Status: Regular flakes and micronized grades are available from regional warehouses for timely fulfillment.
Sample Program: Small quantities (e.g., 500g) are provided for formulation testing and performance evaluation.
Packaging Options:
Industrial grade: 20 kg multi-ply paper/woven bags, 25 kg fiber drums.
Micronized grade: 15 kg or 20 kg fiber drums or paper bags with polyethylene liners.
Custom packaging: Available for bulk orders.
Shipping: Classified as a non-hazardous material; standard freight options include express, less-than-truckload (LTL), and full truckload shipping.
Quality Certifications & Compliance
Manufacturing Standards: Production facilities are ISO 9001:2015 certified, ensuring consistent quality management systems. Pharmaceutical Grade HCO is manufactured in compliance with current Good Manufacturing Practices (cGMP) to meet pharmacopeial requirements .
Batch Documentation: Each shipment is accompanied by a Certificate of Analysis (COA), which comprehensively documents key quality parameters including appearance, melting point, acid value, iodine value, saponification value, hydroxyl value, and particle size (for micronized grades) .
Safety Documentation: Safety Data Sheets (SDS/MSDS) are available in multiple languages to support global regulatory compliance and workplace safety.
Third-Party Validation: Independent test reports (e.g., from SGS) can be provided upon customer request, verifying product quality and compliance with specified standards.
Specialized Certifications: Additional compliance documents are available to meet diverse market requirements, including:
Non-GMO declaration (confirming no genetically modified castor oil sources)
Allergen statement (certifying absence of common allergens)
Halal certificate (for food, cosmetic, and pharmaceutical applications in Muslim-majority markets)
Kosher certificate (aligning with Jewish dietary and product standards)
Sustainable castor oil certification (validating environmentally responsible sourcing and production)
Technical Support & Value-Added Services
Expert Technical Consultation: A dedicated team of application specialists offers online support for product selection, heating/dissolution process optimization, formulation adjustment (e.g., thickening efficiency, gelling performance), and process adaptation to ensure seamless integration into customer workflows .
Customized Solutions: For qualified long-term partners, tailored services are available, including customized particle size specifications, bespoke quality standards aligned with specific application needs, and personalized packaging solutions .
Regulatory Support: Comprehensive documentation packages are provided to facilitate product registration in target markets, streamlining compliance with local regulatory frameworks.
Available Documentation
Certificate of Analysis (COA)
Safety Data Sheet (SDS/MSDS)
Technical Data Sheet (TDS)
Product Specification (PS)
Certificate of Origin (COO)
Halal certificate
Kosher certificate
Sustainable castor oil certification
Pharmaceutical excipient documentation (including Drug Master File (DMF), where applicable)