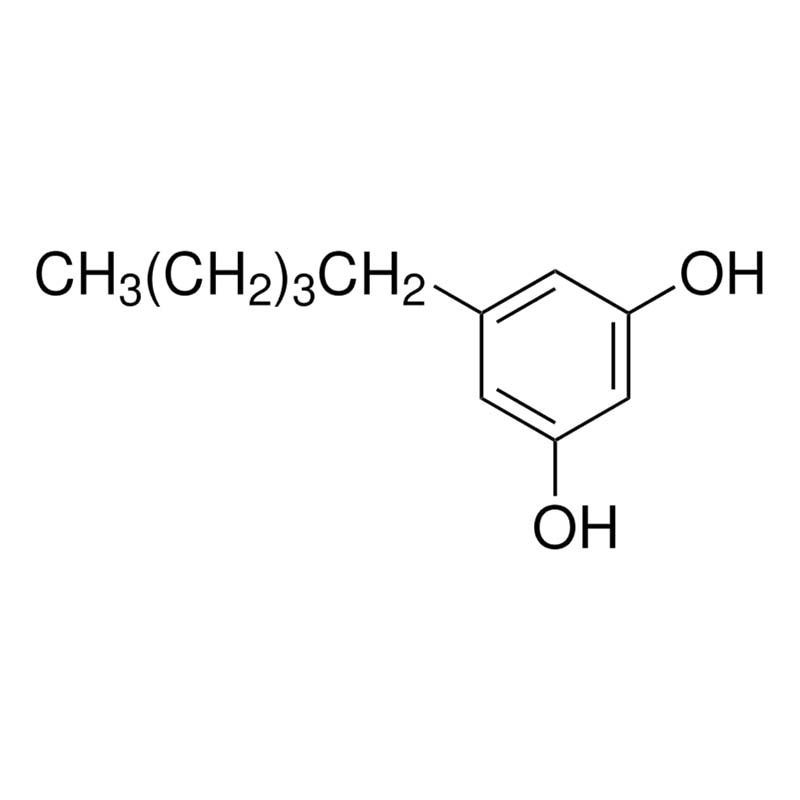
OlivetolCAS:500-66-3
This compound exerts robust fungicidal and bactericidal effects against a broad spectrum of pathogenic microorganisms.
At elevated dosage levels, it promotes DNA cleavage via Cu(II) chloride-mediated oxidative pathways, which occurs under conditions involving molecular oxygen.
Clinically validated data confirms its therapeutic effectiveness against retroviral infections such as HIV. Additionally, it has demonstrated significant inhibitory activity against various malignant tumors and carcinoma cell lines.
Potent Antimicrobial Properties of Olivetol
Olivetol possesses remarkable antimicrobial activity, exerting both fungicidal and bactericidal effects against a wide array of pathogenic microorganisms.
DNA Strand Scission Mechanism at Elevated Concentrations
Modern research has confirmed that at increased dosage levels, this 3,5-dihydroxypentylbenzene-derived compound is capable of inducing DNA strand scission. The underlying mechanism involves Cu(II) chloride-mediated oxidative cleavage pathways, which take place under conditions containing molecular oxygen
Parameters
Melting point | 46-48 °C(lit.) |
Boiling point | 164 °C |
density | 1.068±0.06 g/cm3(Predicted) |
Fp | >230 °F |
storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
solubility | Chloroform (Slightly), Methanol (Slightly) |
form | Solid |
pka | 9.59±0.10(Predicted) |
color | Colourless to Beige |
Stability: | Light Sensitive |
InChI | InChI=1S/C11H16O2/c1-2-3-4-5-9-6-10(12)8-11(13)7-9/h6-8,12-13H,2-5H2,1H3 |
InChIKey | IRMPFYJSHJGOPE-UHFFFAOYSA-N |
SMILES | C1(O)=CC(CCCCC)=CC(O)=C1 |
CAS DataBase Reference | 500-66-3(CAS DataBase Reference) |
NIST Chemistry Reference | 1,3-Benzenediol, 5-pentyl-(500-66-3) |
EPA Substance Registry System | Olivetol (500-66-3) |
Safety Information |
Hazard Codes | Xi |
Risk Statements | 36/37/38 |
Safety Statements | 26-36/39 |
WGK Germany | 3 |
RTECS | VH2880000 |
HS Code | 2907290090 |
Therapeutic Efficacy in Retroviral Infections and Malignancies