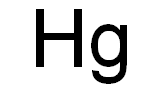Mercury CAS# 7439-97-6
CAS Number:7439-97-6
Chemical Formula: H
Synonyms:
Blue mass
Blue pill
Colloidal mercury
MOQ (Minimum Order Quantity): 1 FCL (Full Container Load)
Appearance: Triple Distilled Liquid
Mercury CAS# 7439-97-6
Mercury is the only metal that is a liquid metal at room temperature. Occurring naturally as mercuric sulfide (in cinnabar ore) because of its low boiling point can be isolated readily from the ore and condensing the resulting vapor to form a metal that is 99.9% pure. Solid mercury is ductile and very malleable and is soft enough to cut with a knife. A poor conductor of heat, mercury can conduct electricity but behaves much like a noble gas. It does not react with most acids but can react with very strong acids and dissolves readily to form amalgams with many metals, including gold, zinc, and aluminum.
Elemental mercury, a silver-white metal, is also known ‘quicksilver’ or ‘hydrargyrum.’ Mercury has been discovered in Egyptian tombs dating as far back as 1500 BC. The chemical symbol, Hg, is derived from the Greek word hydrargyros, meaning ‘water silver.’ Mercury was known in antiquity and used by alchemists. Its neurological effects were recognized early, and its use in the hat-making trade gave rise to the phrase ‘mad as a hatter.’ Mercury has been used commercially and medically for centuries. In the past it was a common constituent of many medications, for example, it was used in the treatment of syphilis. Use of mercury has been drastically reduced in recent years. Within the twentieth century, mercury used to be in every physician’s or pharmacist’s armamentarium, for example, calomel was commonly used in infant teething powders in the 1930s and 1940s.
Application of Mercury
Cadmium is used in electroplating, in nickelcadmiumstorage batteries, as a coating forother metals, in bearing and low-meltingalloys, and as control rods in nuclear reactors.Cadmium compounds have numerousapplications, including dyeing and printingtextiles, as TV phosphors, as pigments andenamels, and in semiconductors and solarcells.
Vegetables and cereals are the main sourcesof dietary Cd while meat and fish contain themetal to a lesser extent.
Amalgams, catalyst, electrical apparatus, cathodes for production of chlorine and caustic soda, instruments (thermometers, barometers, etc.), mercury vapor lamps, extractive metallurgy, mirror coating, arc lamps, boilers, coolant, and neutron absorber in nuclear power plants.
Electrical apparatus; measurement and control systems such as thermometers and sphygmomanometers; agricultural and industrial poisons; catalyst; antifouling paint; dental practice; gold mining.
| Mercury Chemical Properties |
| Melting point | -38.9 °C |
| Boiling point | 356.6 °C(lit.) |
| density | 13.54 |
| vapor density | 7 (vs air) |
| vapor pressure | <0.01 mm Hg ( 20 °C) |
| storage temp. | Poison room |
| solubility | H2O: soluble |
| form | Triple Distilled Liquid |
| Specific Gravity | 13.5 (20/4℃) |
| Odor | Odorless |
| Flame Color | Red |
| resistivity | 95.8 μΩ-cm, 20°C |
| Electrical Conductivity | 1× 106S/m |
| Water Solubility | 20–30μg/L in H2O; soluble in boiling H2SO4, HNO3 [KIR81] [HAW93] |
| Thermal Conductivity | 8.171 W/(m·K) at 25 ℃ |
| Merck | 13,5925 |
| Henry's Law Constant | 1.3×10-3 mol/(m3Pa) at 25℃, Burkholder et al. (2019) |
| Exposure limits | TLV-TWA 0.05 mg/m3 for Hg vapor, and 0.10 mg/m3, as Hg for alkyl mercury and inorganic compounds (ACGIH); ceiling 0.1 mg/m3 (OSHA); IDLH 28 mg/m3 (NIOSH). |
| Dielectric constant | 1.0(148℃) |
| Stability: | Stable. Incompatible with strong acids, sodium thiosulfate, ammonium hydroxide. |
| InChI | 1S/Hg |
| InChIKey | QSHDDOUJBYECFT-UHFFFAOYSA-N |
| SMILES | [Hg] |
| Surface tension | 425.41mN/m at 20°C |
| CAS DataBase Reference | 7439-97-6(CAS DataBase Reference) |
| IARC | 3 (Vol. 58) 1993 |
| EPA Substance Registry System | Mercury (7439-97-6) |
| Safety Information |
| Hazard Codes | T,N,Xn,C,T+ |
| Risk Statements | 25-48/21/22-51/53-50/53-33-23-20/21/22-34-36/37/38-23/24/25-48/23-26-61-52/53-36/38 |
| Safety Statements | 7-45-60-61-36-36/37/39-26-36/37-53 |
| RIDADR | UN 2809 |
| OEL | STEL: 0.1 mg/m3 |
| WGK Germany | 3 |
| RTECS | OV4550000 |
| F | 10 |
| TSCA | TSCA listed |
| HS Code | 2805 40 90 |
| HazardClass | 8 |
| PackingGroup | III |
| Storage Class | 6.1A - Combustible acute toxic Cat. 1 and 2 very toxic hazardous materials |
| Hazard Classifications | Acute Tox. 2 Inhalation Aquatic Acute 1 Aquatic Chronic 1 Repr. 1B STOT RE 1 |
| Hazardous Substances Data | 7439-97-6(Hazardous Substances Data) |
| Toxicity | LCLO inhal (rabbit) 29 mg/m3 (30 h) PEL (OSHA) 0.1 mg/m3 (ceiling) TLV-TWA (ACGIH) 0.025 mg/m3—skin |
| IDLA | 10 mg Hg/m3 |
Fact Factory and Equipment Show