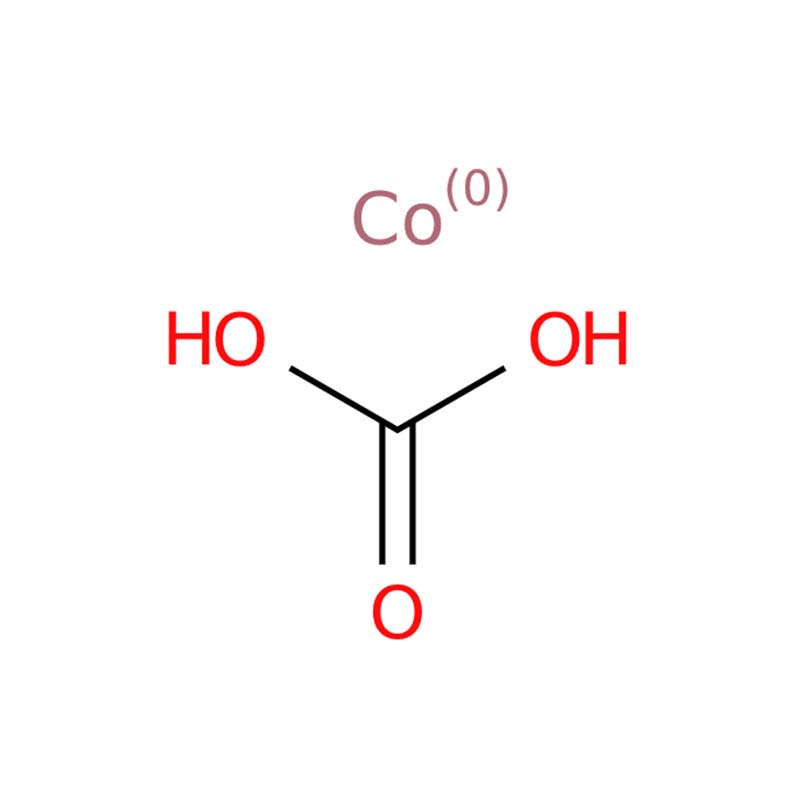
Cobalt Carbonate CAS:513-79-1
Cobalt(II) carbonate (CoCO₃) is a reddish, paramagnetic solid that acts as an intermediate in the hydrometallurgical extraction of cobalt from ores. It serves both as an inorganic pigment and a precursor for catalyst synthesis. Found naturally as the rare mineral spherocobaltite (which has a red-to-pink hue), it is commonly produced industrially by precipitating cobalt sulfate solutions with sodium bicarbonate.
| Category | Specification & Details |
| CAS Number | 513-79-1 |
| Physical Properties | - Appearance: Pink powder- Melting Point: Decomposes (no fixed melting point)- Density: 4.13 g/mL at 25 °C (lit.)- Specific Gravity: 4.13 |
| Solubility | - Water Solubility: Insoluble in water, alcohol, and methyl acetate- Insoluble in ethanol- pKsp: 12.84 |
| Storage & Stability | - Storage Conditions: 2–8 °C, under inert gas (nitrogen/argon)- Stability: Air-sensitive; incompatible with strong oxidizing agents |
| Other Key Data | - Vapor Pressure: 0 Pa at 20 °C- Exposure Limit (ACGIH): TWA 0.02 mg/m³- Merck Index: 14,2436 |
| Hazard Codes | Xn |
| Risk Statements | 22-36/37/38-40-43 |
| Safety Statements | 26-36/37 |
| WGK Germany | 3 |
| TSCA | Yes |
| HazardClass | 9 |
| HS Code | 28369930 |
| Hazardous Substances Data | Cobalt(II) carbonate (513-79-1) |
Cobalt(II) carbonate occurs naturally as the mineral sphaerocobaltite and has a wide range of industrial uses. It is used in ceramic manufacturing, serves as the base for cobalt-based pigments, acts as a catalyst in chemical reactions, and functions as a temperature indicator. Additionally, it is a precursor for various cobalt(II) salts and is used in animal feed supplements to boost the nutritional value of forage for livestock.